Uncovering the secret of insulin growth factor ternary complex
Insulin-like growth factor (IGF) is a hormone that greatly influences growth in fetuses and children, but also body maintenance and metabolism in adults. IGF regulates cell proliferation, differentiation, and survival by activating IGF receptors distributed in cell membranes of various tissues. However, IGF is so unstable that its half-life is less than 10 minutes in its free state.
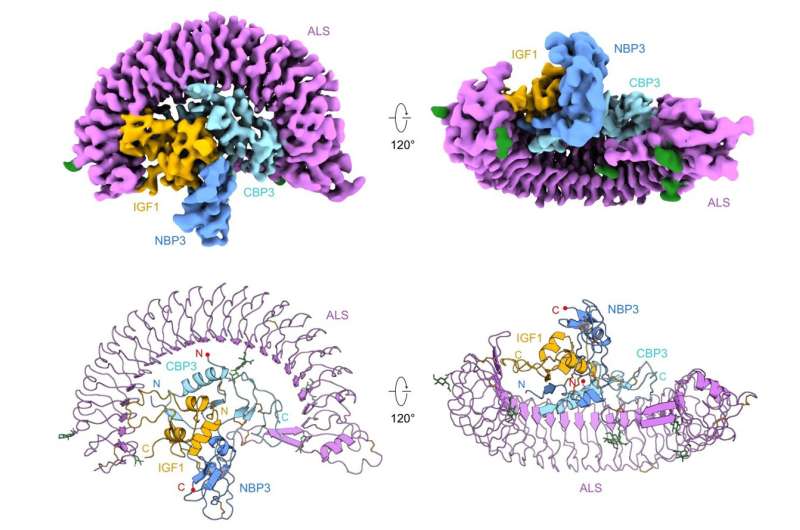
For, this reason more than 70% of IGF circulating in the body is bound to IGFBP (IGF Binding Protein) and ALS (Acid Labile Subunit). This IGF/IGFBP/ALS ternary complex is stable for a much longer duration of 12 hours and therefore serves as a carrier for IGF. In addition, the ternary complex also plays a role in regulating IGF so that it can act only when needed. However, the structure of the IGF/IGFBP/ALS ternary complex has not been well studied, making it difficult to understand how the complex is assembled and how IGF is activated.
Now, a team led by Professor Kim Ho Min at the Center for Biomolecular and Cellular Structure within the Institute for Basic Science (IBS) in Daejeon, South Korea, determined the structure of IGF1/IGFBP3/ALS ternary complex for the first time. It is believed that this structural information could broaden the understanding of growth-related diseases such as growth hormone deficiency and ALS deficiency.
The stable ternary complex was successfully expressed and purified through an animal cell expression system and a series of purification processes. The cryogenic transmission electron microscope (cryo-EM), data hub, and supercomputing resources of IBS were used to obtain its high-resolution three-dimensional molecular structure. It was discovered that IGF1 was surrounded by IGFBP3 to first form a binary complex. Then, ALS once more wraps this binary complex like a parachute, which protects IGF1 from being easily degraded within the body.
Armed with the new knowledge of the protein structure, the researchers were now able to understand the structural basis for many diseases associated with the mutations in these proteins. In particular, many known point mutations in ALS were found to cause disease by inducing misfolding of ALS and hindering the formation of the ternary complex. On the other hand, known point mutations in IGF did not appear to have any negative effects on their ability to form complexes, but rather cause disease by decreasing IGF's affinity to IGF receptors.
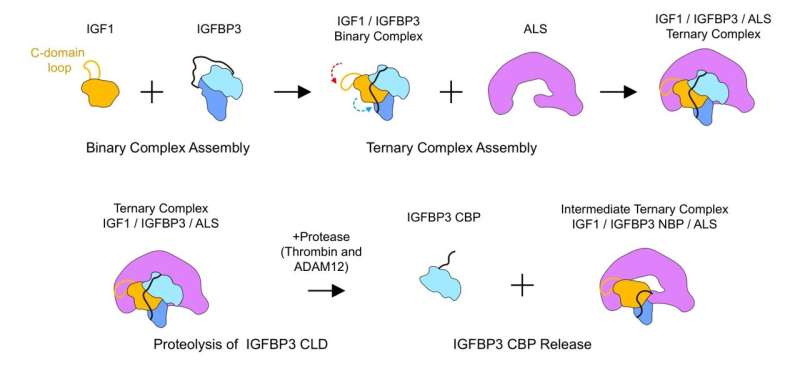
The researchers did not stop there, as they further explored how this ternary complex is assembled. First, the researchers ran SDS-PAGE of ALS, IGFBP3, and IGF1 proteins to verify if the complex is or isn't formed. They also fused GFP (green fluorescent protein) onto the IGF1 protein and used fluorescence detection chromatography to trace the GFP labeled protein as they succeed or fail to form complexes with the other two proteins. From this, they learned that IGFBP3 or IGF1 alone cannot form a binary complex with ALS. Moreover, it was discovered that IGF must first form a binary complex with IGFBP, and only after that, both proteins can bind to ALS to properly assemble a ternary complex.
Furthermore, the researchers modified IGFBP to better determine the role of specific domains of the protein. When the N terminal domain (NBP) was removed, the protein was shown to be incapable of binding to IGF, hinting that the domain is required for the initial binary complex formation. The researchers also substituted the central-linker domain (CLD3) of the IGFBP with different variants. Contrary to NBP, all variants of CLD domains allowed the formation of the binary complex with IGF1. However, it was shown that some CLD variants allowed the modified IGFBP to bind to ALS even without IGF. This means that it is likely that the formation of the initial IGF1/IGFBP3 binary complex allows for some conformational change to the CLD3 domain to facilitate ternary complex formation with ALS.
In addition, the researchers monitored how the complex can be disassembled to make IGF bioavailable. In order to do so, they treated both IGF/IGFBP binary complex and the final ternary complex using a protease called thrombin. After the proteolysis, it was observed that the C-terminus domain (CBP) of IGFBP is released, and an unstable intermediate ternary complex is formed. This process was believed to be an important step in activating the IGF, which is otherwise inactive when it is bound within a complex.
The first author Kim Hyojin says, "The first and foremost step before investigating further processes is to understand the structure. This structure-based approach and proper biochemical assays allowed us to unveil the mechanism of IGF complex formation and activation."
"This new knowledge of molecular structure and activation mechanism of the IGF ternary complex is expected to greatly contribute to the development of therapeutic agents and research related to adolescent growth or IGF-related diseases," explains Professor Kim Ho Min, the corresponding author of the study.
The study is published in Nature Communications.
More information: Hyojin Kim et al, Structural basis for assembly and disassembly of the IGF/IGFBP/ALS ternary complex, Nature Communications (2022). DOI: 10.1038/s41467-022-32214-2
Journal information: Nature Communications
Provided by Institute for Basic Science