Ternary ruthenium complex hydrides can catalyze ammonia synthesis under mild condition

Ammonia is the feedstock for nitrogen fertilizers and thus vital to the sustainable development of society.
Industrial ammonia synthesis catalyzed by Fe or Ru is carried out under high temperature and high pressure with enormous CO2 emission. The development of efficient catalyst enabling ammonia synthesis under mild condition is important.
Recently, a joint research team led by Prof. CHEN Ping from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) and Prof. Tejs Vegge from Technical University of Denmark (DTU) has found that ternary complex hydrides of ruthenium and alkali (alkaline earth) metals (i.e., Li4RuH6 and Ba2RuH6) performed well in catalyzing ammonia synthesis under mild condition.
This study was published in Nature Catalysis on Nov. 18.
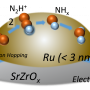
They found that the key to achieve efficient ammonia synthesis under mild condition lay in the unique configuration and function mechanism of the ternary complex hydride center, which was different from the conventional Ru based catalysts.
In ternary hydride catalytic system, N2 underwent a non-dissociative activation over the electron-rich [RuH6] anionic centers through the involvement of hydridic hydrogens in mediating electron and proton transfers and Li or Ba cations in stabilizing NxHy species, leading to ammonia production with superior kinetics.
Hence, the synergistic engagement of all the components of the ternary complex hydrides enabled a reaction path with a narrow energy span, and thus favored ammonia production under mild conditions.
"This work opens an avenue for the design and development of efficient catalysts to tackle the conversion of kinetically stable molecules," said Prof. Chen.
More information: Qianru Wang et al, Ternary ruthenium complex hydrides for ammonia synthesis via the associative mechanism, Nature Catalysis (2021). DOI: 10.1038/s41929-021-00698-8
Journal information: Nature Catalysis
Provided by Chinese Academy of Sciences