New ternary hydrides of lanthanum and yttrium join the ranks of high-temperature superconductors
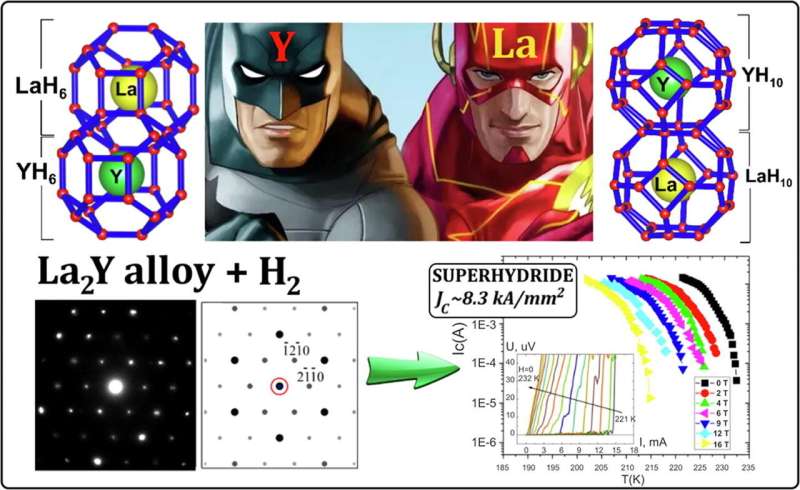
A team led by Skoltech professor Artem R. Oganov studied the structure and properties of ternary hydrides of lanthanum and yttrium and showed that alloying is an effective strategy for stabilizing otherwise unstable phases YH10 and LaH6, expected to be high-temperature superconductors. The research was published in the journal Materials Today.
Cuprates had long remained record-setters for high-temperature superconductivity until H3S was predicted in 2014. This unusual sulfur hydride was estimated to have high-temperature superconductivity at 191–204 K and was later obtained experimentally, setting a new record in superconductivity.
Following this discovery, many scientists turned to superhydrides, which are abnormally rich in hydrogen, and discovered new compounds that became superconducting at even higher temperatures: LaH10 (predicted and then experimentally shown to have superconductivity at 250-260 K at 2 million atmospheres) and YH10 (predicted to be an even higher temperature superconductor). Despite the similarity between yttrium and lanthanum, YH10 proved to be unstable, and thus far no one has succeeded in synthesizing it in its pure form. Having reached the upper limit of critical temperatures for binary hydrides, chemists turned to ternary hydrides which appear as the most promising path towards still higher temperature superconductivity. Finally in 2020, after over 100 years of research, scientists were able to synthesize the first room-temperature superconductor—a ternary sulfur and carbon hydride − with a critical temperature of +15 degrees Celsius.
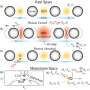
In their recent work, scientists from Skoltech, the Institute of Crystallography of RAS, and V.L. Ginzburg Center for High-Temperature Superconductivity and Quantum Materials studied ternary hydrides of lanthanum and yttrium with different ratios of these two elements.
"Although lanthanum and yttrium are similar, their hydrides are different: YH6 and LaH10 do exist, while LaH6 and YH10 do not. We found that both structures could be stabilized by adding the other element. For example, LaH6 can be made more stable by adding 30 percent of yttrium, and its critical superconductivity temperature is slightly higher as compared to YH6," professor Oganov says.
In addition, the research has helped to elucidate the general profile of superconductivity in ternary hydrides. "We realized that ternary and quaternary hydrides have progressively less ordered structures and a much greater width of the superconducting transition than binary hydrides. Also, they require more intensive and longer laser heating than their binary counterparts," lead author and Skoltech Ph.D. student Dmitrii Semenok explains.
The scientists believe that the study of ternary hydrides holds much promise for stabilizing unstable compounds and enhancing their superconducting performance.
More information: Dmitrii V. Semenok et al, Superconductivity at 253 K in lanthanum–yttrium ternary hydrides, Materials Today (2021). DOI: 10.1016/j.mattod.2021.03.025
Journal information: Materials Today
Provided by Skolkovo Institute of Science and Technology