Potassium hunting on protein factories

Groundbreaking research at the new long-wavelength macromolecular crystallography beamline (I23) at Diamond Light Source has for the first time demonstrated the location of potassium ions in bacterial ribosomes. Ribosomes are the protein factories of cells and although they are vital for life, little was known of the sites of metal ions that are crucial for their structure and function. The work recently published in Nature Communications showcases the fantastic applications of the I23 beamline and sheds light on the important role of potassium ions.
Ribosomes are giant protein factories that reside in cells from all forms of life, and are responsible for the accurate conversion of genetic information into proteins. They are the most complex RNA-protein assemblies in the cell and need metal ions to maintain their structure and function. Although such an important constituent of a cell, the exact type and location of metal ions within these large complexes were yet to be defined. In fact, earlier efforts to characterise the ribosome overemphasised the importance of magnesium ions, so other metal ions were largely ignored.
An international team of researchers sought to fully define the role of potassium within ribosomes. Using the unique long-wavelength macromolecular crystallography beamline (I23) at Diamond, the team was able to pinpoint hundreds of potassium ions within bacterial ribosomes. The cutting-edge technique demonstrated for the first time on a 3-D structural basis that potassium ions were not only involved in the overall formation of the structure of ribosomal RNA (rRNA) and ribosomal proteins, but that they were also playing an important role in its function.
These results fill a huge knowledge gap and could also lead to potential therapeutic applications. By fully understanding the intricacies of bacterial ribosomes, it is hoped that they might be targeted to develop new classes of antibiotics.
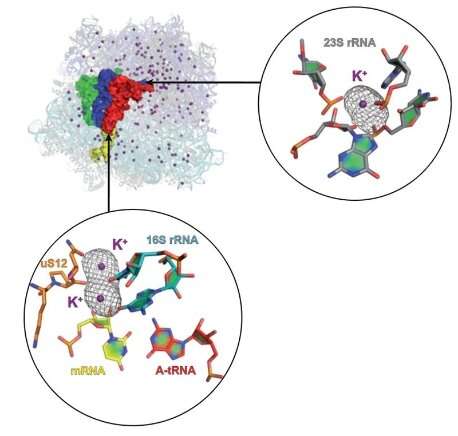
Magnesium by default
Ribosomes are essential for protein synthesis and are composed of two complex subunits. Although their structure has previously been thoroughly characterised by X-ray crystallography and cryo-electron microscopy (cryo-EM), the exact metal ions that support their structure and function have eluded scientists.
When ribosomes are subjected to diffraction experiments, the metal ions contribute to regions of electron density. By default, researchers have attributed this density to magnesium ions to generate 3-D structural models. However, it is known that potassium also plays an important role within ribosomes as its withdrawal leads to unfolding.
This European collaborative effort involves Gulnara Yusupova and Marat Yusupov from the Institute of Genetics and Molecular and Cellular Biology (IGBMC, Strasbourg, France), and Alexey Rozov from a spin-out company from the IGBMC called "RiboStruct. "We have been working for 30 years towards our main goal, to understand how the atomic structure of the ribosome ultimately determines its extraordinary function: protein synthesis. Potassium is a most abundant cation in the cells, and as such was deemed important for all cellular processes, but its influence on translation was never studied in detail." the team of Gulnara Yusupova and Marat Yusupov stated. Thus they embarked on a lengthy study aided by Dr. Armin Wagner's team at I23 to fully characterise potassium ions within bacterial ribosomes.

Unique facility
Taking over two years to develop, I23 is a unique facility at Diamond that covers an energy range not available at any other synchrotron in the world. Dr. Wagner, Principal Beamline Scientist at I23 and one of the authors of the study explained the capabilities of the beamline: "The unique wavelength range allows us to really target the binding of potassium. So we can do experiments around the absorption edge that allow us to calculate 3-D maps which highlight the atoms contributing to the scattering."
The study focussed on bacterial ribosomes from Thermus thermophilus. The researchers concentrated their efforts on crystal structures of the ribosomes in complex with messenger RNA and transfer RNA. By measuring the anomalous scattering signals at the K-edge they could detect the potassium ions.
Dr. Wagner's team at I23 helped to plan and perform the study, which was the first of its kind in the world. Studying one of the most complex biological systems was taxing and the teams from IGBMC and RiboStruct spent over a year analysing the data to locate potassium ions with unprecedented precision.
Vital role of potassium
The team saw hundreds of potassium ions, and many were in important positions within the ribosome. The most important one stabilises the decoding centre when bound to the messenger RNA, which conveys genetic information to the ribosome. These insights really show the vital role of potassium in protein synthesis.
Typically, cryo-EM is largely used to explore ribosomal structure, but this study demonstrates that crystallography can offer unique insights. The accurate location of potassium and indeed other metal ions is not possible with electrons yet.
The teams will continue their efforts into characterising ribosomes using both cryo-EM and crystallography and they intend to look at higher organisms in the future. At Diamond, Dr. Wagner aims to realise the potential of I23 as a valuable research tool. "I23 is currently the only facility where this research could have been carried out. It shows that I23 can determine the position of light atoms like potassium, calcium, chlorine, phosphorus, sulphur, all atoms of high biological significance. This study is just the start and we are expecting more exciting results from the beamline," he concluded.
More information: Alexey Rozov et al. Importance of potassium ions for ribosome structure and function revealed by long-wavelength X-ray diffraction, Nature Communications (2019). DOI: 10.1038/s41467-019-10409-4
Journal information: Nature Communications
Provided by Diamond Light Source




















