Microtubules anchored to proteins in the nuclear membrane position muscle cell nuclei
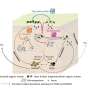
Scientists at A*STAR have revealed how microtubules, part of a cell's 'skeleton," position the multiple nuclei in muscle cells. Malfunctions in this crucial developmental process are linked to muscular dystrophies, a group of debilitating diseases that cause progressive weakness and loss of muscle mass.
Brian Burke of A*STAR's Institute of Medical Biology was drawn to the topic more by accident than by design. His research focuses on the genes that encode proteins that are active in the "nuclear envelope," a membrane around the cell nucleus. In muscle cells, the nuclear envelope contains proteins usually associated with the centrosome, a cytoplasmic structure which organizes the microtubules, but for years this remained a niche subject. "It was just an odd phenomenon that no-one working in the centrosome field understood the significance of, and no one outside the field was aware of," he says.
Yin Loon Lee, who studied the centrosome during his Ph.D. before joining Burke's lab, brought the topic to his attention. Burke's recent research had revealed that protein pairs in the nuclear envelope, known as LINC complexes, play a role in positioning the nucleus in many cells. "It seemed like a good bet that these [complexes] might be involved in anchoring centrosomal proteins at the muscle cell nucleus," explains Burke.
To test this idea, Lee and Burke fished out proteins that interact with the LINC protein Nesprin-1 in early- and late-stage muscle cells. Comparing the two lists revealed 446 proteins that interact with Nesprin-1 specifically in late-stage muscle cells, including four centrosomal proteins. Meanwhile, researchers in France had performed an RNAi screen to identify genes involved in microtubule nucleation at the nuclear envelope. "The only candidate they came up with was Nesprin-1," says Burke. The teams decided to collaborate to solve the mystery.
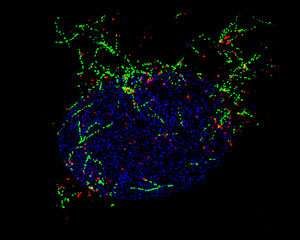
Lee engineered cells lacking either Nesprin-1 or its LINC partners. In these cells, three of the four centrosomal proteins remained in the cytoplasm rather than accumulating at the nuclear envelope. The same mislocalization was apparent in cells from patients with a congenital muscular dystrophy caused by a Nesprin-1 mutation. By depleting each of the three centrosomal proteins individually, the team discovered which protein was required for microtubule formation at the nuclear envelope.
In muscle cells with a missing or defective copy of Nesprin-1, the nuclei clumped together rather than spreading out. Understanding how Nesprin-1 and centrosomal proteins position the nuclei of muscle cells may help clarify the development of muscular dystrophies.
More information: Petra Gimpel et al. Nesprin-1α-Dependent Microtubule Nucleation from the Nuclear Envelope via Akap450 Is Necessary for Nuclear Positioning in Muscle Cells, Current Biology (2017). DOI: 10.1016/j.cub.2017.08.031
Journal information: Current Biology