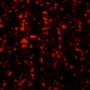
Formation of cell membrane component domains in artificial lipid bilayer

Research conducted by Toyohashi University of Technology in collaboration with Tohoku University elucidated the fusion process of proteoliposomes with an artificial lipid bilayer and the mechanism behind this process. In addition, the researchers also discovered that the domains composed of all cell membrane components exist as "islands" isolated from the artificial membrane. These findings will lead to further understanding of the functions of membrane proteins, which are an important target of drug development, as well as the development of experimental techniques. The results of this research were published in Scientific Reports on December 20, 2017.
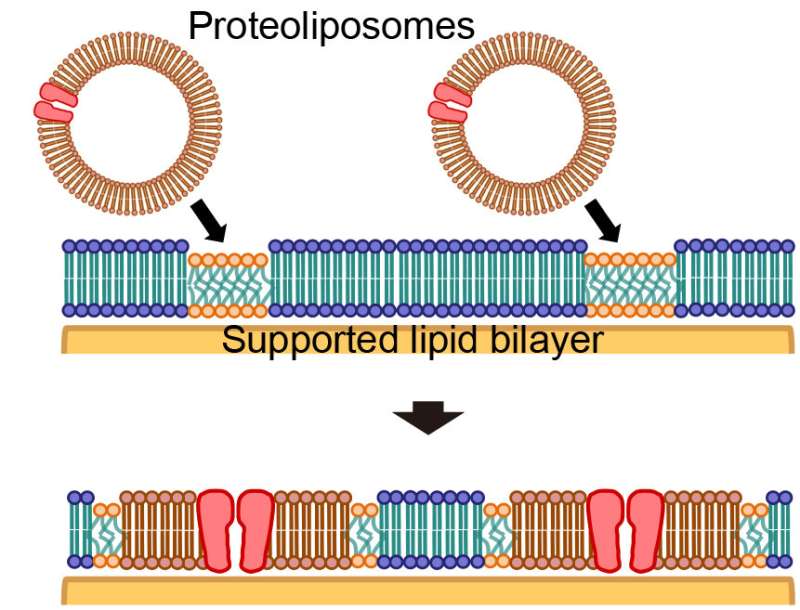
All exchanges of materials, signals, and energy in and out of cells to maintain biological activity are performed through membrane proteins and lipids on the cell membrane. Since these processes have a strong influence on neurotransmission and metabolism, they are important research targets in the fields of biology, medicine and drug development. Components of the cell membrane including membrane proteins and lipids are generally derived from cultured cells, and spherical structures of lipid bilayer membranes including these derived proteins are called proteoliposomes.
Because membrane proteins maintain their structure and functions by remaining within a lipid bilayer, artificial lipid bilayers are commonly used for measuring the functions of membrane proteins without affecting their activity. After the fusion of proteoliposomes with an artificial lipid bilayer, the cell membrane environment must be maintained; experimental conditions for this fusion have been derived through accumulated empirical evidence.
The research group led by Ryugo Tero, associate professor at Toyohashi University of Technology in collaboration with Tohoku University, discovered that "islands" made of cell membrane components grow within an artificial lipid bilayer through observation of the fusion of proteoliposomes derived from cultured cells with an artificial lipid bilayer. Furthermore, they also found that the artificial lipid bilayer and proteoliposomes do not mix, and that membrane proteins and lipids inside the cell membrane formed isolated domains away from the artificial lipid bilayer. The size and distribution of these "islands" were found to be dependent on the type of cells that the proteoliposomes were derived from. In addition, they also clarified that microdomains (domains with a specific composition of lipids) serve as a specific site for the fusion of proteoliposomes.
Associate professor Ryugo Tero says, "We were very surprised when we saw the spreading of dark islands made of cell membrane components in a sea of the bright artificial lipid bilayer labeled with fluorescence. The phosphatidylcholine, phosphatidylethanolamine and cholesterol used in this study to make the artificial lipid bilayer are major components in the cell membrane. Although proteoliposomes also contain these same lipid components, it was very strange to find that they did not mix with each other. This result provides very valuable information in that the cell membrane components are not being mixed into the surroundings and dispersed, but form clusters in the artificial lipid bilayer. By using this experimental technique, for example, we could also observe the phenomenon of collaborative interaction between multiple proteins and lipids in the cell membrane."
Professor Ayumi Hirano-Iwata at Tohoku University says that "In our study of ion channels, the most important factor affecting the success rate of measurements is whether proteoliposomes fuse with an artificial lipid bilayer or not. We had been searching for the right experimental conditions each time we changed the type of cells or membrane proteins. By understanding the membrane fusion process and its mechanism as clarified by this study, the efficiency of our experiments will be greatly improved."
The research group believes that the fusion process of proteoliposomes and its mechanism elucidated by this research will accelerate the research of ion channels and membrane proteins which are important targets of drug development. In addition, the "islands" made of cell membrane components will provide useful information for understanding complex biological reactions in which multiple proteins and lipids are involved, as well as for developing high-throughput membrane protein screening technology.
More information: Ryugo Tero et al, Formation of Cell Membrane Component Domains in Artificial Lipid Bilayer, Scientific Reports (2017). DOI: 10.1038/s41598-017-18242-9
Journal information: Scientific Reports
Provided by Toyohashi University of Technology