New microscopy technique allows mapping protein synthesis in living tissues and animals
Researchers at Columbia University have made a significant step toward visualizing complex protein metabolism in living systems with high resolution and minimum disturbance, a longstanding goal in the scientific community. In a recent study published in ACS Chemical Biology, Assistant Professor of Chemistry Wei Min's research team has reported a light microscopy method to image where the new proteins are produced and where the old proteins are degraded inside living tissues and animals.
Proteins carry most biological functions in life. Biologists have long learnt that new proteins are produced and old ones are destroyed over time, as required by the metabolic activity of living organisms. Many complex physiological and disease processes involve protein synthesis and degradation in space and time. For example, formation of long-term memory requires activity-dependent protein synthesis in specific neurons while the devastating Huntington's disease often disrupts protein degradation pathways of affected cells.
Technically, however, it is very challenging to visualize protein metabolism in a noninvasive and global (i.e., proteome) manner especially for live systems. Previous methods using radioactive amino acids have to work with dead samples. Imaging mass spectrometry is invasive, thereby not compatible with live systems. Fluorescence based techniques using unnatural amino acids generally require non-physiological fixation of cells.
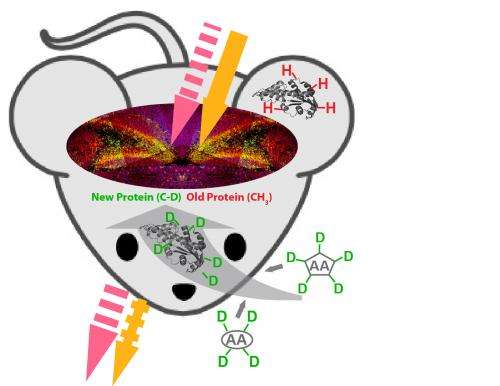
To address this problem, Min and his team exploited a novel combination of chemical labeling and physical detection. Specifically, they coupled an emerging laser-based technique called stimulated Raman scattering (SRS) microscopy with metabolic labeling of deuterated amino acids (D-AAs) by cell's native protein synthesis machineries. In the specially prepared D-AAs, the normal hydrogen atoms are replaced by deuterium atoms, which exhibit almost identical physical-chemical properties to those of hydrogen except for their heavier masses. When the carbon-deuterium bond stretches, it produces a Raman scattering signal at a unique "frequency" (different from natural molecules inside cells). The newly synthesized proteins, built upon the incorporated D-AAs, can then be specifically detected by SRS through the unique vibrational signature from carbon-deuterium bonds. Using this method, Min and his team have demonstrated imaging newly synthesized protein in live cells, as reported in an earlier publication Proc. Natl. Acad. Sci. USA. 110, 11226, 2013.
"The new ACS Chem Biol paper has significantly expanded the concept of coupling SRS with metabolic labeling of D-AAs," said lead author Lu Wei, a Ph.D. candidate in chemistry. Besides monitoring protein synthesis, this technique can also probe protein degradation, by tracking the signal decay of the methyl group (CH3) attributed to pre-existing proteins. Moreover, inspired by the classic pulse-chase analysis of complex protein dynamics, two-color pulse-chase imaging was accomplished by rationally dividing the whole D-AAs collection into two structurally different sub-sets with distinct vibrational signatures. In this way, Min's team was able to probe progression dynamics of aggregate formation of mutant huntingtin proteins in live cells.
"Equipped with the much improved technique, we can pursue biological applications well beyond the earlier demonstration on simple cell lines," Wei said. In this new study, the team imaged the spatial distribution of newly synthesized proteins inside live brain tissue slices with pronounced signal observed in dentate gyrus, suggesting high metabolic activity in this region. The intricate relationship between protein synthesis and neuronal plasticity is currently under investigation on this platform.
"The major advantage of our technique lies in its non-toxicity and minimal invasiveness, as administration of D-AAs in the model organisms appears to be non-toxic even for a long duration," Wei added. The team has mapped newly synthesized protein in living animals including zebrafish and mice. Strong signals are detected in the somites at the embryonic zebrafish tail and the liver and intestine tissues from examined mice. All these new demonstrations support the technique for in vivo interrogation.
"In addition to basic research, our technique could also contribute greatly to translational applications," said Min. "Considering that stable isotope labeling and SRS imaging are both compatible with live humans, we envision a bright prospect of applying this platform to performing diagnostic and therapeutic imaging in humans."
More information: ACS Chemical Biology, pubs.acs.org/doi/abs/10.1021/cb500787b
Journal information: ACS Chemical Biology
Provided by Columbia University