Lethal weapon: bacteria's high-risk suicide strategy
(Phys.org)—New research shows how some bacterial cells keep a 'suicide complex' ready to hand at all times.
Research published today in the journal Proceedings of the National Academy of Sciences shows that some bacterial cells carry a molecular 'suicide complex' to kill themselves in the event of lethal infection by viral parasites. Such 'altruistic suicide' prevents or limits viral replication and protects the rest of the bacterial population from subsequent infection.
The new research demonstrates that bacteria accomplish this through a high-risk strategy in which their lethal weapon is kept to hand at all times, but is neutralised until viral infection of the bacterial cell triggers its release from inhibition. In the longer term, the discovery could be exploited to enable the development of new small molecule antibacterial drugs.
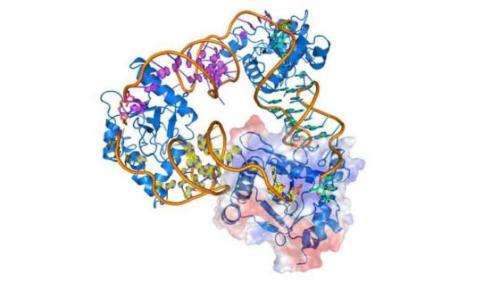
The mechanism was discovered in the bacterial plant pathogen Pectobacterium atrosepticum by researchers led by Professors George Salmond and Ben Luisi in the University of Cambridge's Department of Biochemistry. Their work shows that a suicide complex, ToxIN, is not induced but exists all the time in the bacterial cell; to avoid killing the bacterial cell, it is held in a suppressed, inert form until viral infection triggers the release of a protein toxin (ToxN) from an RNA antitoxin (ToxI) partner. The toxin then causes the death of both the bacterium and the infecting virus.
The success of the antiviral system therefore depends heavily on maintaining a very strong inhibition or suppression of the toxin by its RNA antitoxin, to ensure that the host cell is not damaged in the absence of invading viruses or other stresses. Small RNAs have multiple essential roles in bacteria, but examples of naturally occurring RNA molecules that act as direct protein inhibitors are rare.
Professor George Salmond, deputy head at the Department of Biochemistry, said: "The results present a picture of ToxIN as an addictive, self-assembling – and potentially lethal – molecular machine, which can drive remarkable adaptive advantages in populations of bacterial hosts, including those under threat from lethal viral predation."
The research, which was funded by the Biotechnology and Biological Sciences Research Council (BBSRC), explores the powerful ToxN-inhibiting activity of the ToxI RNA. It shows that the ToxI RNA 'neutralises' its toxin partner through the self-assembly of a triangular ToxI-ToxN macromolecular complex, previously observed by earlier BBSRC-funded crystallographic studies published in Nature Structural and Molecular Biology in 2011.
The assembly of the inhibited complex is driven entirely by the sequence of the ToxI RNA, and its interactions with ToxN. The study shows that ToxI RNAs are highly selective inhibitors, each active against only their own specific toxin partner. The structure of a second ToxI-ToxN complex, encoded by a plasmid (a small piece of circular DNA that is separate from the single chromosome of the bacterial cell) of the bacterium Bacillus thuringiensis reveals that this selectivity is a consequence of subtle, complementary structural variations in both RNA and protein – the precise molecular recognition needed to form an inactive complex cannot occur between mismatched partners.
In addition, the work shows that ToxIN systems promote their own maintenance on plasmids as selfish DNA that probably increases their spread, and retention, in bacterial populations.
Journal information: Proceedings of the National Academy of Sciences , Nature Structural and Molecular Biology
Provided by University of Cambridge