This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Unlocking the secrets of cell behavior on soft substrates: A paradigm shift in mechanobiology
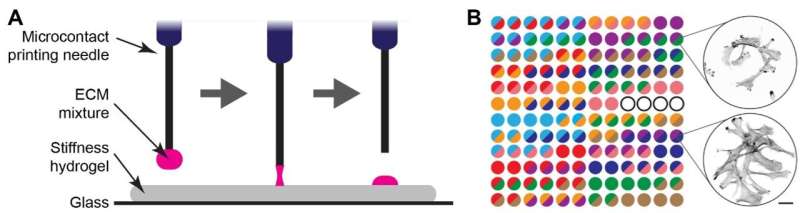
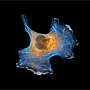
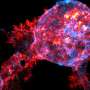
A research group from the University of Turku and Turku Bioscience Center together with Misvik Biology Ltd in Finland have developed a new method for studying how cancer cells function in softer and stiffer tissue environments. This insight challenges the existing paradigm, opening up new possibilities for research in cancer biology and tissue engineering. The results of the study have been published in PNAS.
A longstanding belief has been that cells outside the body prefer to spread and grow on stiffer surfaces. This is similar to when we walk on a concrete sidewalk (very stiff) and find it preferable to walking in mud (very soft). For this reason, cells, including stem cells, are continuously cultured on very stiff plastic or glass for research purposes.
This idea also resonates with cancer cells thriving within a hard lump they form in tissues. Usually, the stiffer the tumor, the poorer the patients' prognosis. However, the stiffness of the tissues in our body (e.g., bone versus brain) is not the same. In fact, some cells like neurons and fat cells grow and function effectively in very soft surroundings.
The research group from the University of Turku and Turku Bioscience Center collaborated with Misvik Biology Ltd, a biotechnology company based in Turku, Finland, to understand how cells function in softer environments and how these could be better modeled outside the human body. They used computational modeling and a large array of growth conditions to meticulously compare cell behavior on soft and stiff surfaces at an unprecedented resolution.
New perspective on an old idea
Using an automated machine, the researchers microprinted different protein mixtures onto soft and stiff surfaces. The proteins chosen were those that typically surround cells in the body and give texture and information about the tissue environment.
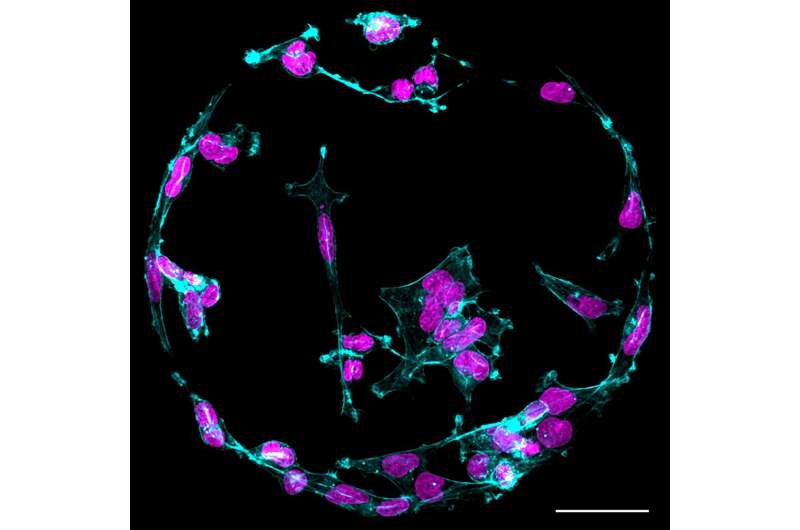
"By using more diverse protein mixtures in cell culture, we begin to get closer to a more physiologically relevant setting outside the body with which we can more effectively model diseased and healthy states," says Dr. James Conway from the InFLAMES research flagship, the lead researcher of the Turku Bioscience team.
"We had a specific system for microcontact printing of different protein mixtures onto culture plates and decided to see how we could expand the system. Once the researchers at Turku Bioscience had the idea, it was straightforward for us to then use the system on different stiffness hydrogels," explains CEO Juha Rantala from Misvik, a key collaborator on the project.
By working together, the team demonstrated that the right combination of proteins can support cells on a soft surface, providing crucial survival cues that result in cell growth similar to that observed on a stiffer surface.
"These findings have unveiled a fundamental new perspective in mechanobiology. We have identified a mechanism through which we can explain how cells can effectively adhere, function, and signal on soft substrates. This insight challenges the existing paradigm, opening up new possibilities for research in cancer biology and tissue engineering," notes Prof. Johanna Ivaska, the principal investigator of the project.
More information: James R. W. Conway et al, Defined extracellular matrix compositions support stiffness-insensitive cell spreading and adhesion signaling, Proceedings of the National Academy of Sciences (2023). DOI: 10.1073/pnas.2304288120
Journal information: Proceedings of the National Academy of Sciences
Provided by University of Turku