This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers uncover NSMF protein's role in relieving DNA replication stress
A team of researchers from the Department of Biological Sciences at UNIST has achieved a significant breakthrough in understanding how brain proteins can help alleviate complications arising from DNA replication stress. This discovery holds immense potential for advancing treatments for various diseases, including cancer, neurological disorders, and age-related conditions that result from disruptions in DNA replication.
Led by Professor Jayil Lee, Professor Jang Hyun Choi, and Professor Hongtae Kim, this collaborative effort has unveiled crucial insights into the intricate processes involving NSMF proteins when confronted with DNA replication stress—a fundamental aspect of cellular functioning.
N-methyl-D-aspartate receptor synaptonuclear signaling and neuronal migration factor (NSMF), a neuronal protein associated with Kallmann syndrome, plays a novel role in neuronal development, regulation of movement, reproductive hormone secretion, and olfactory perception. Dysfunctions in this protein can lead to rare conditions like Kallmann syndrome—an affliction characterized by impaired reproductive function and loss of sense of smell.
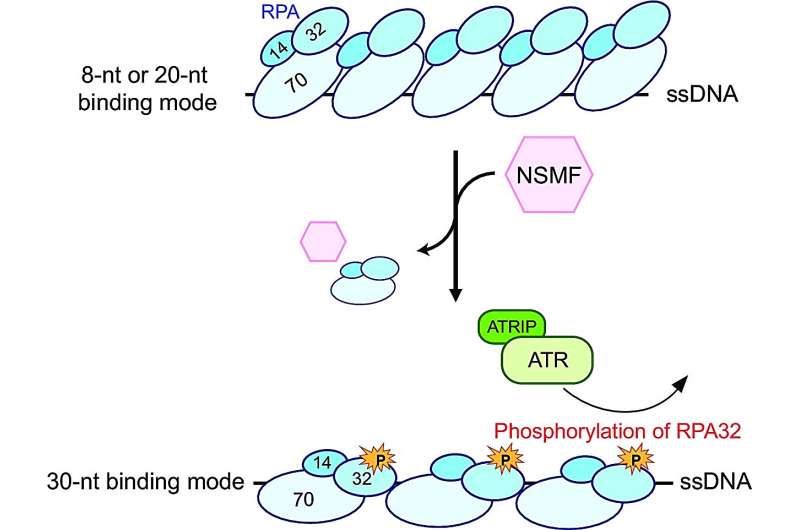
In their study focused on alleviating DNA replication stress using NSMF proteins, the research team observed that when protein synthesis encounters obstacles during replication due to various factors causing stress on DNA structure—resulting in single-stranded regions—the replication protein A (RPA) binds uniquely to these exposed single strands.
The combined RPA molecules then undergo phosphorylation—a chemical process involving attachment of phosphate groups composed of phosphorus and oxygen. Phosphorylated RPA recruits other proteins that help alleviate replication stress at specific sites along the DNA strand—restoring normal activity.
Interestingly, RPA exhibits weak or strong binding affinity towards single-stranded regions during its interaction with DNA. Through their investigation involving NSMF proteins, the research team discovered that NSMF selectively displaces some weakly bound RPAs while promoting a transition into more stable binding modes for the remaining RPAs.
This shift favors an enhanced phosphorylation process, catalyzed by the ataxia telangiectasia and RAD3-related (ATR) kinase—an enzyme crucial for DNA damage response. The team's findings demonstrate that this mechanism accelerates the relief of replication stress.
"This study has significant potential to impact treatments related to cancer, neurological disorders, and age-related conditions by unraveling molecular mechanisms associated with DNA replication," commented Professor Lee.
"Considering NSMF's close relationship with Kallmann syndrome, we anticipate its contribution to advancements in treating this disease," added Yujin Kang, the first author of the study.
The study findings have been published in Nucleic Acids Research.
More information: Yujin Kang et al, Alteration of replication protein A binding mode on single-stranded DNA by NSMF potentiates RPA phosphorylation by ATR kinase, Nucleic Acids Research (2023). DOI: 10.1093/nar/gkad543
Journal information: Nucleic Acids Research