This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Mikado in the cell: Arrangement of proteins could be responsible for diseases
Processes inside human cells are tightly regulated in time and space by various enzymes and proteins. However, if processes become unbalanced—for example, because cells experience increased stress—these processes can also lead to diseases.
For example, proteins can "aggregate"—that is, cluster together and form extended, ordered, straight fibers, similar to a Mikado. While most proteins have a well-defined three-dimensional structure, some exist in cells without any structure, like a long string.
This category of proteins is called intrinsically disordered. Recently such intrinsically disordered proteins have received considerable attention as driving cellular organization and have been linked to neurodegeneration. However, it is unclear how these disordered and flexible proteins become structured to build the Mikado.
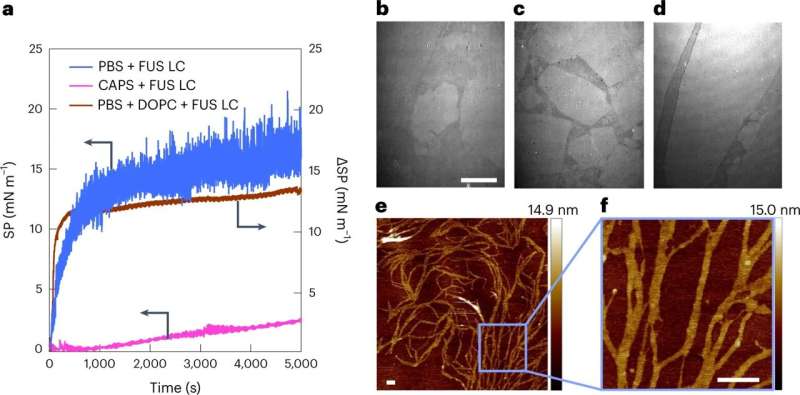
A team of researchers at the Max Planck Institute for Polymer Research and the University of Texas has now shown that interfaces can trigger aggregation of a model intrinsically disordered protein—called FUS (fused in sarcoma). This disordered protein is flexible in the bulk, but at a hydrophobic interface, it forms fibers. These FUS proteins form connected "Mikado-networks " that cannot be easily broken down and may contribute to the development of neurodegenerative diseases.
"We looked at the formation of FUS fibers at the hydrophobic interface using laser-based methods including spectroscopy and microscopy," say Mischa Bonn and Yuki Nagata. The researchers observed the formation of fibers through the collective ordered assembly of intrinsically disordered proteins. The researchers further showed that protein mobility was dramatically reduced upon fiber formation: the proteins are stuck in the fibers they form.
"We were able to demonstrate that hydrophobic interfaces—for example, small oily droplets in cells—can seed molecular ordering and fiber formation," explains Sapun Parekh, also a group leader in Mischa Bonn's department and a professor at the University of Austin, Texas. "This formation happens at surprisingly low concentrations: concentrations 600 times lower than necessary for forming loose protein clusters in solution," Parekh adds.
The scientists hope their research will contribute to future understanding of how neurodegenerative diseases develop. They have now published their study in Nature Chemistry
More information: Daria Maltseva et al, Fibril formation and ordering of disordered FUS LC driven by hydrophobic interactions, Nature Chemistry (2023). DOI: 10.1038/s41557-023-01221-1
Journal information: Nature Chemistry
Provided by Max Planck Society