Scientists identify interactions that stabilize a neurodegeneration-associated protein

Most of the well-studied proteins in our bodies are like metal; some can change shape easily, like aluminum foil, and others are rigid, like steel beams, but they typically have a solid, well-defined structure. Many other essential proteins are more like water—able to change phase from liquid to solid ice.
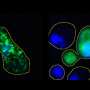
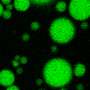
One of these important phase-changing proteins is called FUS. In healthy cells, FUS switches between floating diffusely and condensing into liquid droplets with other proteins to make, edit and deliver the blueprints for protein production. However, FUS also has a "solid" or aggregate phase that has been found in some people with severe cases of amyotrophic lateral sclerosis (ALS) and a type of dementia called frontotemporal dementia.
A team of researchers led by Nicolas Fawzi, an associate professor in the Department of Molecular Pharmacology, Physiology and Biotechnology at Brown University, used a combination of techniques to determine the atomic interactions that stabilize the liquid, yet "condensed" phase of FUS.
"What we want to understand is the atomic details of these interactions so that we know what kind of treatments would be important for ALS and other diseases," said Fawzi, a co-corresponding author on the paper who is also affiliated with the Carney Institute for Brain Science. "First we need to know the structural differences between the normal form and the disease form, so we know where to put in a wrench to stop it. We can't design a drug to bind to something when we don't know what it looks like."
The findings were published on Monday, July 1, in the journal Nature Structural and Molecular Biology.
Fawzi said you can think of the condensed liquid droplets FUS can form within cells as somewhat like the condensation that forms on a cold glass on a humid day. Both the water droplets and the humid air contain water molecules, but they are in distinct phases.
In addition to its implication in ALS—commonly called Lou Gehrig's disease—the disordered region of FUS is known to be associated with some types of cancer including Ewing's sarcoma, Fawzi said. In fact, the acronym FUS stands for FUsed in Sarcoma.
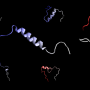
Using a combination of nuclear magnetic resonance (NMR) spectroscopy, Raman spectroscopy and computational modeling, Fawzi's team including Brown doctoral student Anastasia Murthy, the lead author on the study, found that the interactions between a disordered region of FUS in the condensed liquid droplets are quite varied and dynamic, he said. FUS does not form any traditional structural elements. However, multiple atomic interactions—including those formed by specific amino acids within the protein, namely glutamine and tyrosine—maintain the condensed yet disordered nature of FUS.
The knowledge of these molecular interactions—which are distinct from the interactions in the neurodegeneration-associated aggregate phase—might someday be used to guide the development of therapeutics that hinder disease-associated aggregation or support the normal condensed phase interactions.
Additionally, the combination of techniques Fawzi's team used to uncover FUS's atomic interactions could be used by other scientists studying intrinsically disordered proteins that also form liquid or solid condensed forms, such as Huntington's, Parkinson's, prion diseases and Type II diabetes in addition to ALS.
"We did these NMR experiments in a new way that allowed us to explicitly look at which atoms in one protein are interacting with atoms in another FUS protein," Fawzi said. "I expect that people will start doing experiments like this because it provides more assurance and detail on the contacts between intrinsically disordered proteins. Disordered proteins do all kinds of important things, and we don't really know how they normally work. When they go wrong, we really don't know what's going on."
Fawzi plans to continue studying FUS. Specifically, he wants to study the entire protein, not just the intrinsically disordered region that was the primary focus of this paper, in test tubes as well as in living cells. He also plans to apply this combination of techniques to continue studying the molecular interactions of other neurodegeneration-associated disordered proteins such as hnRNPA2 and TDP-43.
More information: Molecular interactions underlying liquid−liquid phase separation of the FUS low-complexity domain, Nature Structural and Molecular Biology (2019). DOI: 10.1038/s41594-019-0250-x , nature.com/articles/s41594-019-0250-x
Journal information: Nature Structural and Molecular Biology
Provided by Brown University