Green hydrogen: Faster progress with modern X-ray sources
Green hydrogen is an energy carrier with a future. It is obtained by electrolytically splitting water with energy from wind or sun and stores this energy in chemical form. To make the splitting of water molecules easier (and to reduce the energy input), the electrodes are coated with catalytically active materials.
Dr. Marcel Risch and his Young Investigator Group "Oxygen Evolution Mechanism Engineering" are investigating oxygen evolution in the electrocatalysis of water. Oxygen evolution in particular must run more efficiently for economical hydrogen production.
Exciting class of materials
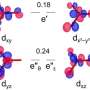
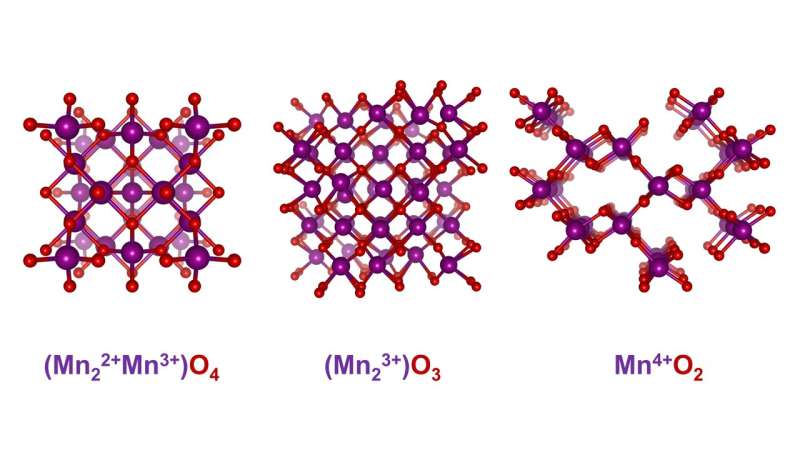
An exciting class of materials for electrocatalysts are manganese oxides, which occur in many different structural variants. "A decisive criterion for suitability as an electrocatalyst is the oxidation number of the material and how it changes in the course of the reaction," explains Risch.
In the case of manganese oxides, there is also a great diversity in possible oxidation states. X-ray absorption spectroscopy (XAS) provides information about the oxidation states: X-ray quanta with suitable energy excite electrons on the innermost shells, which absorb these quanta. Depending on the oxidation number, this absorption can be observed at different excitation energies. Risch's team has constructed an electrolysis cell that enables XAS measurements during electrolysis.
X-ray absorption spectroscopy
"With X-ray absorption spectroscopy, we can not only determine the oxidation numbers, but also observe corrosion processes or phase changes in the material," says Risch.
Combined with electrochemical measurements, the measurement data thus provide a much better understanding of the material during electrocatalysis. However, the required high intensity of the X-rays is only available at modern synchrotron light sources. In Berlin, HZB operates BESSY II for this purpose. There are about 50 such light sources for research worldwide.
Time scales from short to long
Risch still sees great potential for the application of X-ray absorption spectroscopy, especially with regard to the time scales of observation. This is because typical measurement times are a few minutes per measurement. Electrocatalytic reactions, however, take place on shorter time scales.
"If we could watch electrocatalysis as it happens, we could better understand important details," says Risch. With this knowledge, cheap and environmentally friendly catalysts could be developed more quickly. On the other hand, many "aging" processes take place within weeks or months. "We could, for example, examine the same sample again and again at regular intervals to understand these processes," Risch advises. This would also make it possible to develop electrocatalysts with long term stability.
The research was published in Angewandte Chemie.
More information: Marcel Risch et al, What X‐Ray Absorption Spectroscopy Can Tell Us About the Active State of Earth‐Abundant Electrocatalysts for the Oxygen Evolution Reaction, Angewandte Chemie (2022). DOI: 10.1002/ange.202211949
Journal information: Angewandte Chemie
Provided by Helmholtz Association of German Research Centres