A catalyst for the development of carbon-neutral technology of the radiation accelerator

The importance of "carbon neutrality" is growing more than ever, as climate change caused by global warming threatens even the human right to live. The Republic of Korea has declared "carbon neutrality by 2050" and is exerting efforts to reduce greenhouse gas emissions. In order to realize carbon neutrality, along with green hydrogen production that reduces the generation of carbon dioxide, CCU technology that utilizes already-generated carbon dioxide is essential.
In order for these two technologies to be effective in reducing greenhouse gas emissions, the energy used must be reduced by increasing the activity of the water oxidation electrode, which induces an electrochemical reaction. For this purpose, attempts have been made to understand the electronic structure of the surface of the catalyst while the reaction continues. However, due to the difficulty in conducting an experiment in an ultra-high vacuum (UHV) condition, it was only indirectly estimated through computational calculations.
At the Korea Institute of Science and Technology (KIST), Dr. Hyung-Suk Oh and Dr. Woong Hee Lee from the Clean Energy Research Center and Dr. Keun Hwa Chae from the Advanced Analysis and Data Center developed a soft X-ray based absorption spectroscopy based on a radiation accelerator (10D XAS KIST beamline) for the first time in Korea. KIST announced that this research has developed a new strategy to fabricate electrodes by observing and analyzing the surface electronic structure during the reaction of the water oxidation electrode applied to "hydrogen production and conversion of carbon dioxide."
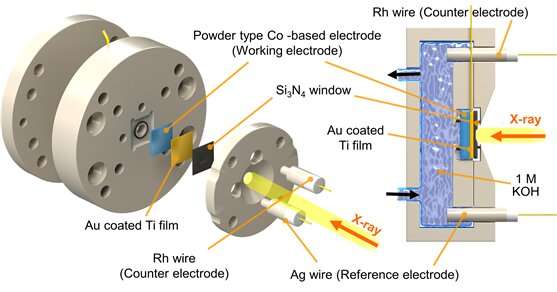
The research team found that general cobalt was reconstructed during the reaction, through measuring the electronic structure and spin states of the electrode surface by using accelerator-based soft X-ray absorption spectroscopy under the UHV condition. Thermodynamically, cobalt is prone to be in a tetravalent oxidation state under oxidation conditions, and its water oxidation activity is very low. It is necessary to maintain a trivalent oxidation state in order to maintain high water oxidation activity, that the process developed by the research team enables to obtain the 3.2 oxidation state and high activity. The developed electrode has a 1,000 times larger electrochemical surface area compared to a commercial cobalt electrode, and 10 times the hydrogen production performance when applied to an actual water electrolysis system.

Dr. Oh says that "by developing an operando soft X-ray absorption spectrometry based on a radiation accelerator, we have taken one step further in understanding the properties of catalyst materials and improving their performance. This is an essential technology for artificial photosynthesis, and is expected to be of great help in improving the performance of the water oxidation electrode, which is an important technology for green hydrogen production and electrochemical reconstruction."
The research was published in Nature Communications.
More information: Woong Hee Lee et al, Electrode reconstruction strategy for oxygen evolution reaction: maintaining Fe-CoOOH phase with intermediate-spin state during electrolysis, Nature Communications (2022). DOI: 10.1038/s41467-022-28260-5
Journal information: Nature Communications
Provided by National Research Council of Science & Technology