Using light to restore cell function
New research from the University of Cincinnati shows early indications that light can be used as a treatment for certain diseases, including cancer.
Researchers from UC, the University of Illinois Urbana-Champaign and the University at Buffalo published the results of their study demonstrating light-activated proteins can help normalize dysfunction within cells in the journal Nature Communications July 25.
Research findings
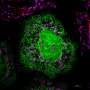
The research centers on the functions of mitochondria, organelles within a cell that act as the cell's "power plant" and source of energy. Organelles are tiny specialized structures that perform various jobs inside cells.
Jiajie Diao, Ph.D., one of the study's authors, said hundreds of mitochondria are constantly coming together (a process called fusion) and dividing into smaller parts (a process called fission) to stay balanced in healthy cells. But when mitochondria are not functioning properly, there is an imbalance of this process of fission and fusion.
This imbalance can lead to a number of mitochondrial diseases, including neurodegenerative diseases like dementia and certain cancers.
Diao said previous research found that another organelle within cells called a lysosome can play a role in mitochondria fission. When a mitochondria comes in contact with a lysosome, the lysosome can act like a pair of scissors and cut the mitochondria into smaller pieces.
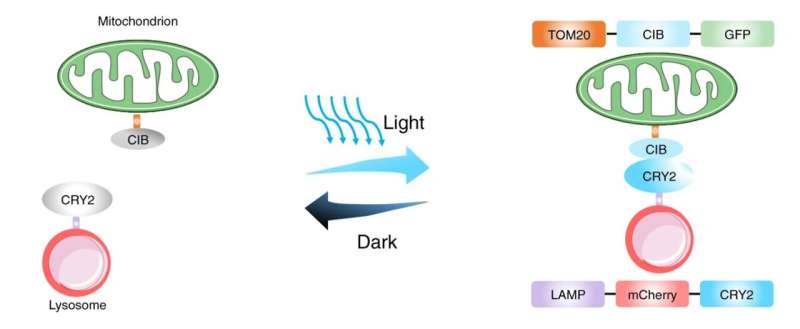
The current research focused on jump-starting the fission process by bringing the lysosomes and mitochondria together within cells. This was accomplished using a technique known as optogenetics, which can precisely control specific cell functions using light.
"Many proteins in plants are light sensitive, informing plants whether it is day or night. Optogenetics borrows these light-sensitive proteins from plants and uses them in animal cells," said Kai Zhang, Ph.D., associate professor at the University of Illinois Urbana-Champaign and study co-author, who developed the optogenetic tools for controlling mitochondria and lysosomes with blue light. "By attaching such proteins to organelles, one can use light to control the interaction between them, such as mitochondria and lysosomes shown in this work," he said.
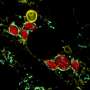
The researchers attached two separate proteins to mitochondria and lysosomes within stem cells. When stimulated by blue light, the proteins naturally bind to each other to form one new protein, which also brings the mitochondria and lysosome into contact. Once they are brought together, the lysosome can cut the mitochondria, achieving fission.
"We found that it can recover the mitochondrial function," said Diao, associate professor in the Department of Cancer Biology in UC's College of Medicine and a University of Cincinnati Cancer Center member. "Some of the cells can even go back to normal. This proves that by just using some simple light stimulation we can at least partially recover the mitochondrial function of the cell."
Diao said this technique could be especially useful for patients with dramatically oversized mitochondria that need to be divided into smaller pieces to achieve normal cell function. The technique could also be aimed at cancer cells, continually separating the mitochondria into smaller and smaller pieces until they can no longer function.
"Eventually the cancer cells will be killed because mitochondria is their energy," Diao said. "Without normal functional mitochondria, all of the cancer cells will be killed."
Since the proteins are activated by light, Diao said it allows for a more targeted approach to specific cells. Only cells exposed to the light are affected, meaning healthy cells nearby do not have their mitochondria unbalanced through the technique.
There are currently other processes that can be used to induce mitochondrial fission, but Diao said the optogenetic method is safer since it does not involve any chemicals or toxic agents.
"What we have is actually the natural process, we're just making it faster," Diao said. "So it's not like a chemical or a therapy or a radiotherapy where you need to reduce the side effects."
Next steps
Diao said his team is already at work on using the same technique to encourage fusion to address issues when mitochondria are unbalanced because they are too small and not coming together as they should within cells.
Further research from Zhang's lab will also include developing new optogenetic systems working with different colors of light, including green, red and infrared, since a longer wavelength will be needed to penetrate human tissue.
"We would like to further expand the toolbox by introducing multicolor optogenetic systems to give us multiple ways to control how organelles behave and interact," Zhang said. "For instance, one color makes organelles come together, while the other color forces them apart. This way, we can precisely control their interactions."
From the current research using human stem cells, the team hopes to progress to test its efficacy using animal models on the way to eventually test the technique in humans through clinical trials. At the same time, Diao said other research groups are studying the use of magnetic fields and acoustic vibrations instead of light to accomplish similar results.
More information: Kangqiang Qiu et al, Light-activated mitochondrial fission through optogenetic control of mitochondria-lysosome contacts, Nature Communications (2022). DOI: 10.1038/s41467-022-31970-5
Journal information: Nature Communications
Provided by University of Cincinnati