Chromosomal errors that develop early lead to embryonic loss in assisted reproductive technology

Assisted reproductive technology, known as ART, is the umbrella term to describe fertility treatments that involve eggs and embryos. Many people turn to ART techniques like in vitro fertilization to resolve fertility problems, but this is an expensive process that can have a low success rate.
In a paper published in June in Biochemical and Biophysical Research Communications, scientists outline a new imaging technique tested in cattle to identify and understand the relationship between chromosomal errors that develop early and embryonic loss.
Previous research in mice has shown a relationship between chromosome errors that happen early in a zygote's development leading to embryonic loss, but this relationship is not well understood in non-rodents. Cattle is an ideal model and next step for ART research because ART is frequently used in cattle breeding and a high rate of embryonic losses is common. There are also several similarities between cow and human embryos and how they develop.
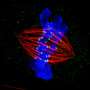
"Chromosome aneuploidy is thought to occur due to chromosome segregation errors that develop early, which may cause embryonic loss," said researcher Satoshi Sugimura of the Institute of Agriculture at the Tokyo University of Agriculture and Technology in Tokyo, Japan. "We developed minimally invasive live-cell imaging and showed the relationship between chromosome segregation error and in vitro and post-transfer embryonic development."
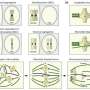
Chromosome segregation happens after fertilization when the zygote begins to go through cell division. When the cell divides, both cells should have a complete set of chromosomes. When this does not happen as it should, it is called a chromosome segregation error. These errors lead to aneuploidy, which is when there are too many or too few chromosomes in a cell. Frequent aneuploidy has been reported in humans and cattle in the early cleavage stages, but its relationship to subsequent development has not been clarified in non-rodents. So, researchers tried to clarify the relationship using live-cell imaging with injection of mRNA encoding histone H2B-mCherry.
"We produced phenotypically normal non-transgenic offspring from bovine embryos with live-cell imaging of chromosome segregation," said Sugimura, marking the birth of the world's first healthy calf from an embryo whose chromosome distribution was observed by the live-cell imaging technique.
"Chromosome segregation error, especially micronucleus formation during early cleavage division was a potential hallmark of preimplantation embryonic loss in non-rodent mammals," said Sugimura. Researchers suspect that specific errors that occur earlier in the cleavage process, such as during the first three mitotic divisions, are more detrimental. These errors, which result in micronuclei formation, are considered severe. Zygotes with abnormal chromosomal segregation during the first three mitotic divisions rarely develop to the blastocyst stage. However, if they do develop into blastocysts, these embryos have the same birth rate as those that did not have abnormal chromosomal segregation.
Half of the calves born during this study came from a group that showed evidence of chromosome segregation errors during early cleavage. Because of this, the error may not be a predictor of postimplantation embryonic loss.
"This technique has shown potential for understanding the relationship between chromosome segregation error and subsequent embryo development, low ART success, and for selecting viable ART-derived embryos for medical and livestock production. In the future, if the mechanism of appearance and origin of chromosome segregation error in early cleavage division can be elucidated and controlled, ART success may be improved," said Sugimura.
More information: Tatsuma Yao et al, Micronucleus formation during early cleavage division is a potential hallmark of preimplantation embryonic loss in cattle, Biochemical and Biophysical Research Communications (2022). DOI: 10.1016/j.bbrc.2022.05.075
Journal information: Biochemical and Biophysical Research Communications
Provided by Tokyo University of Agriculture and Technology