Structure of key protein for cell division puzzles researchers
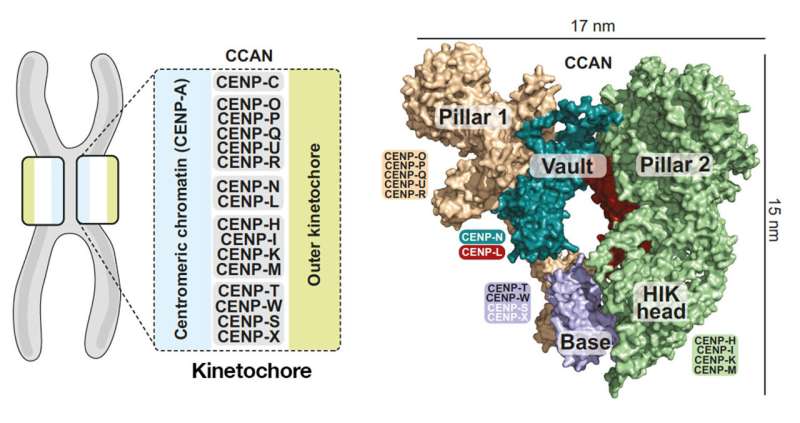
Human cell division involves hundreds of proteins at its core. Knowing the 3D structure of these proteins is pivotal to understand how our genetic material is duplicated and passed through generations. The groups of Andrea Musacchio and Stefan Raunser at the Max Planck Institute of Molecular Physiology in Dortmund are now able to reveal the first detailed structure of a key protein complex for human cell division known as CCAN. By using cryo-electron microscopy, the researchers show important features of the complex's 16 components and challenge previous assumptions about how the complex is able to recognize the centromere, a crucial region of chromosomes in cell division.
At the center of cell division
The centromere is a constriction in the chromosome, made of DNA and proteins. Most importantly, the centromere is the dock for the kinetochore, a machinery of about 100 proteins that drives the separation of two identical chromosomes during cell division and their delivery to the daughter cells. Previous research has shown that the kinetochore docks onto the centromere through the CCAN complex: The CCAN interacts with the centromere protein A, the landmark protein of the centromere. CCAN is also responsible for replenishing the centromere protein A once the cell division has taken place. Yet, the details of the interaction between CCAN and the centromere protein A remain elusive.
"Understanding how CCAN recognizes and binds to the centromere could potentially lead us to build a centromere from scratch," says Musacchio. The centromere is a major hurdle for synthetic biologists who aim to engineer artificial chromosomes to restore missing functions or introduce new ones in cells.
Unresolved questions at the core
Scientists identified the CCAN complex over 15 years ago. "Yet, building up a pipeline to synthesize all proteins in vitro has been a major obstacle," says Musacchio. After obtaining a first reconstitution of the human CCAN complex in vitro, Musacchio joined forces with Stefan Raunser, also at MPI Dortmund, who applied cryo-electron microscopy on the whole CCAN protein complex.
In the new publication, the MPI groups have been able to determine the 3D structural details of the human CCAN complex, highlighting its unique features and the implications for an interaction with the centromere protein A.
"Contrary to what was expected, this structure does not directly recognize the centromere protein A in the standard configuration," says Musacchio.
The centromere protein A is most commonly packed with DNA and other proteins as a nucleosome, the standard unit of the genetic material. The authors are now suggesting that the centromere protein A may be embedded in the centromere with a different configuration that may facilitate the crucial interaction with CCAN. They plan to identify conditions that could lead to this new configuration and prove their hypothesis.
The research was published in Molecular Cell.
More information: Marion E. Pesenti et al, Structure of the human inner kinetochore CCAN complex and its significance for human centromere organization, Molecular Cell (2022). DOI: 10.1016/j.molcel.2022.04.027
Journal information: Molecular Cell
Provided by Max Planck Institute of Molecular Physiology