New insights into an ancient protein complex
Cells rely on membranes to protect themselves from the outside world. But cell membranes can't be fully closed because nutrients and other molecules have to be able to pass through. To achieve this, cell membranes have many types of channels and pores. Also, there are receptors embedded in the membrane that continuously monitor the outside world and signal to the cell interior. Extensive collaboration between five VIB groups resulted in a better understanding of the machinery that plants use to regulate the protein composition of their outer membrane. This discovery, published in Science Advances, enhances our basic knowledge of how the plasma membrane composition can be adapted based on external stimuli, an essential process for life on earth.
The molecular architecture of TPLATE
Complex life has complex cells, also known as eukaryotic cells. Unlike bacteria, for example, the cells of complex life have many distinct internal compartments known as organelles. These organelles exchange material among themselves. To do so, the organelles have a few tricks, one of which is vesicle trafficking. This means that they use a part of their own membrane as a bag for the goods to be exchanged.
A recent discovery showed that plants heavily rely on a protein complex named the TPLATE complex to do so. This complex is not only present in plants, but also in a wide range of other eukaryotes, which suggests it is evolutionarily old and part of a protein complex family of which all other members are intensively studied. However, because this particular complex is not present in the most-studied model organisms (animals and yeasts), its existence and function remained under the radar for a very long time.
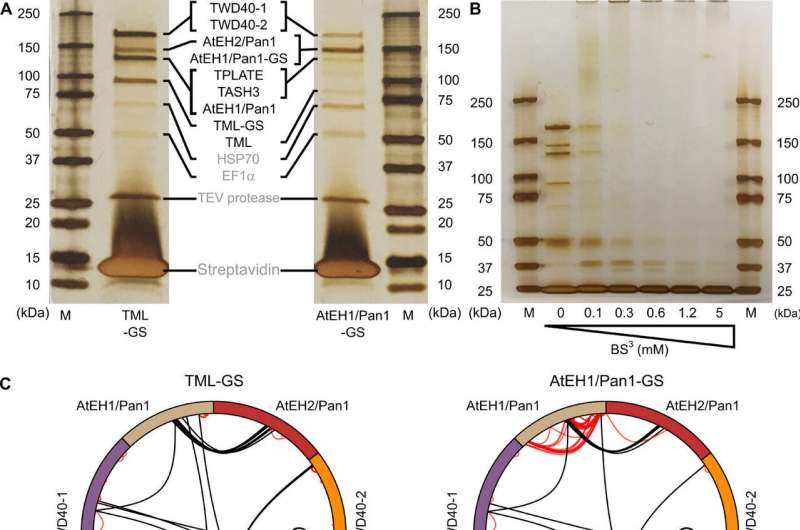
In this study, VIB teams have reveal TPLATE's molecular architecture for the first time. They achieved this by crosslinking mass spectrometry and computer simulations. These new insights revealed the orientation of this complex toward the membrane as well as the delicate relationship between the different domains of its subunits.
These findings are important to increase the knowledge of crucial eukaryotic processes. Indeed, the structure of this complex now allows us to compare it with known structures of its close relatives that are present in all eukaryotes, including animals and yeasts, allowing researchers to visualize the evolution of these trafficking complexes.
To obtain both structural and functional insight into this enigmatic complex required an integrative, collaborative approach. Five VIB research groups and one group from the Czech Republic contributed within their specific expertise to perform experiments ranging from lipid-binding studies to structural biology approaches.
The novel structural insight was mostly generated based on crosslinking mass spectrometry, performed with the help of the VIB Proteomics core facility.
"A major benefit of working at VIB is that it greatly encourages and facilitates access to knowledge and expertise that allows research groups to successfully embark on joint projects that lie far beyond their comfort zone," says Prof. Daniel Van Damme.
This study will form the foundation of further scientific work and will open doors for the generation of novel and safer herbicides or modulation of stress responses in plants.
More information: Klaas Yperman et al. Molecular architecture of the endocytic TPLATE complex, Science Advances (2021). DOI: 10.1126/sciadv.abe7999
Journal information: Science Advances
Provided by VIB (the Flanders Institute for Biotechnology)