Organoids reveal inflammatory processes in chlamydia infections
For a long time, researchers were only able to examine human cells infected with bacteria by using cancer cell lines. However, these transformed cells often give a false impression of the infection process. Fallopian tube organoids from normal human fallopian tube cells, on the other hand, reflect the natural structure of the tissue. Scientists from the Max Planck Institute for Infection Biology in Berlin have now investigated fallopian tube cells chronically infected with Chlamydia for many months. The results show that the cells react to the bacterial infection with increased production of LIF—an inflammatory messenger associated with ectopic pregnancies. In addition, the bacteria cause persistent epigenetic changes to the DNA.
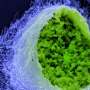
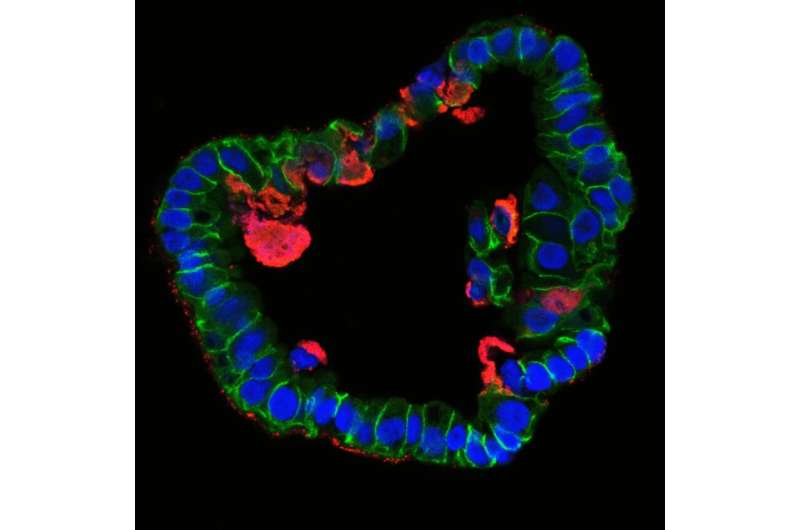
Epithelia form a barrier between the outside world and internal organs and are therefore the first line of defence against pathogens. Today, scientists can use stem cells to cultivate epithelial cell organoids, i.e. cell clusters that replicate natural tissues. This enables them to investigate what happens in healthy human cells during infections without having to employ cancer cells. "Although the development of organoids is time-consuming and costly, their use has led to a quantum leap in our understanding of Chlamydia infections," says Thomas Meyer, Director at the Max Planck Institute for Infection Biology.
Meyer's research group has selected the widespread pathogen Chlamydia trachomatis for their study, which often does not cause any symptoms when sexually transmitted and therefore goes unnoticed. This favours the development of chronic infections, which in women sometimes ascend into the fallopian tubes and are associated with infertility, ectopic pregnancy and ovarian cancer. Because Chlamydia infections are extremely common—more than 100 million new infections per year are estimated worldwide—such serious consequences are a cause for concern.
Alternative to HeLa-cells
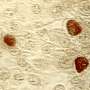
Up to now, scientists have barely been able to investigate the consequences of an infection because fallopian tubes are very difficult to access for clinical investigations. In addition, for a long time there was no model system in which they could analyse the natural course of an infection. "In the past, we were dependent on HeLa cells—a human cell line cultivated from cervical cancer—for infection experiments with Chlamydia. However, these experiments lasted a maximum of 72 hours, after which all cells died," explains Mirjana Kessler, head of the study. "However, when we first looked at time-lapse images of infected fallopian organoids, the difference was astonishing." The organoids stay alive for weeks and months, while the bacteria infect cells and create new offspring, which in turn infect new cells.
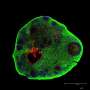
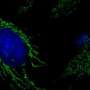
The team suspects that the difference is mainly due to the fact that organoids resemble the natural structure of the epithelium: Their cells are not flat like cancer cells, but tightly packed together and three-dimensional, with a distinct inner and outer surface. This enables them to fend off infections more effectively. Only a subset of cells is infected and subsequently pushed out of the epithelium by the surrounding cells, thus preventing tissue damage. "The cells and bacteria achieve a kind of equilibrium that is very similar to a real human infection. This allows infected organoids to survive for months," adds Thomas Meyer. "This is the first time we have succeeded in reproducing the course of a chronic bacterial infection in mucosal cells."
Epigenetic changes
The organoids react to Chlamydia with increased production of LIF, an inflammatory messenger associated with ectopic pregnancies. This is the first concrete indication of a possible mechanism underlying this complication. In addition, the team has observed changes in DNA that occur as cells age. These changes are permanent and persist even after the bacteria have been successfully eradicated. Such epigenetic long-term changes could explain how Chlamydia infections might promote the development of cancer many years later.
More information: Chronic Chlamydia infection in human organoids increases stemness and promotes age-dependent CpG methylation. Nature Communications; 18 March, 2019. DOI: 10.1038/s41467-019-09144-7
Journal information: Nature Communications
Provided by Max Planck Society