Scientists successfully obtain synthetic growth factor compatible with the native protein
Growth factors are ligands that play important roles in the body—they are responsible for tissue regeneration, wound healing, and the maintenance and growth of cells. Stimulating growth factor-dependent pathways is therefore an important therapeutic strategy to promote the regeneration of tissues due to injuries and diseases. Using synthetic substitutes of growth factors is the primary method for artificially stimulating the desired pathways. However, synthetic substitutes are often not as efficient and comprehensive in inducing the required biological changes. Kunio Matsumoto at Kanazawa University and colleagues have recently created a substitute for one such growth factor, that produces a comparable biological response.
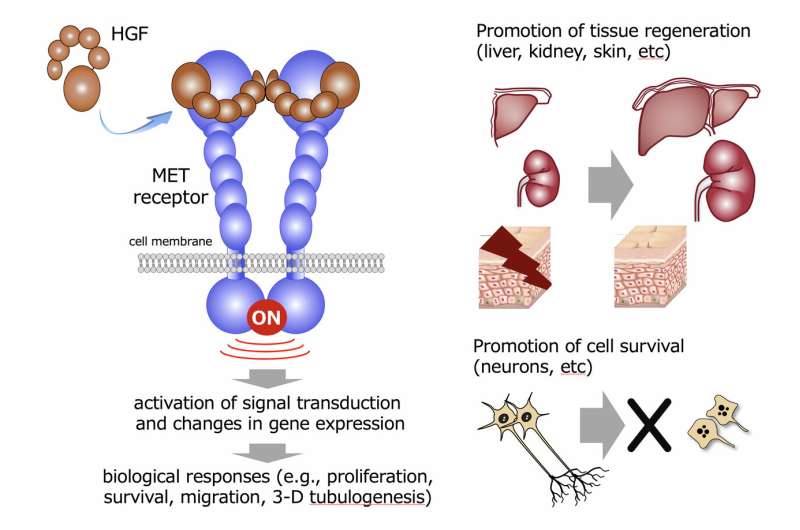
The hepatocyte growth factor (HGF) binds to and activates a receptor known as MET (Fig. 1). In other words, HGF is a native ligand for the MET receptor. The MET receptor is a transmembrane protein. Upon extracellular binding of HGF to the extracellular region of MET, pairing of the receptors (called 'dimerization') occurs, which allows the intracellular chemical change of the receptors (called 'phosphorylation'). This receptor phosphorylation means receptor activation, which triggers activation of intracellular biological signal transduction pathways, leading to a variety of biological responses in cells.
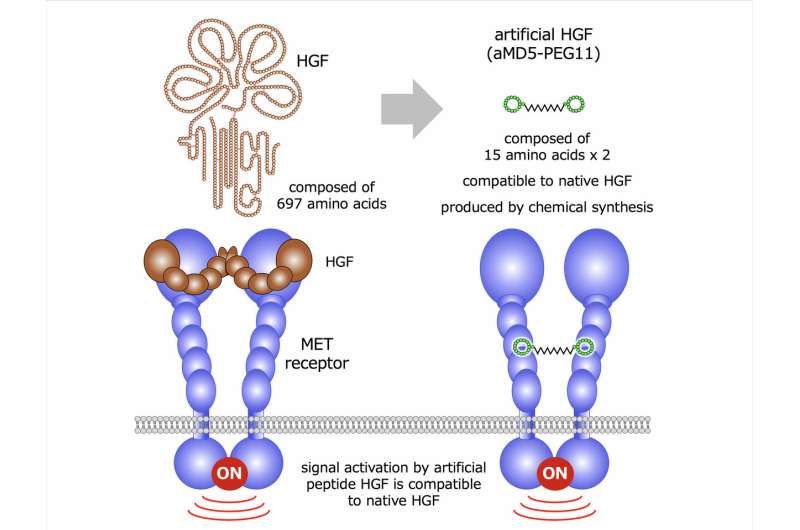
To mimic the actions of HGF, the researchers created a macrocyclic peptide molecule (aMD5-PEG11) that activates the MET receptor (main fig). This molecule is composed of two chemically cross-linked macrocyclic peptides. Such cyclic molecules are flexible in structure and can bind and activate MET. Furthermore, aMD5-PEG11 docks at a different region of MET than HGF; aMD-PEG11 and HGF therefore did not directly compete for MET and nullify each other's effects. Whether aMD-PEG11 also induced these structural changes was then assessed in two different human cell types. aMD-PEG11 induced the same extent of coupling/dimerization and phosphorylation of the MET receptor in a comparable ability to HGF.
If the structural changes induced by HGF and aMD5-PEG11 were similar, but were the biological effects similar, too? MET activation by HGF induces migration of cells. This migration was also observed by aMD-PEG11. MET activation by HGF also results in activation of other signaling proteins such as AKT and ERK. aMD-PEG11 also had comparable effects on the activation of these molecules. These effects were canceled when a specific inhibitor for MET was added, indicating that these effects were definitely results of MET activation induced by aMD-PEG11 or HGF.
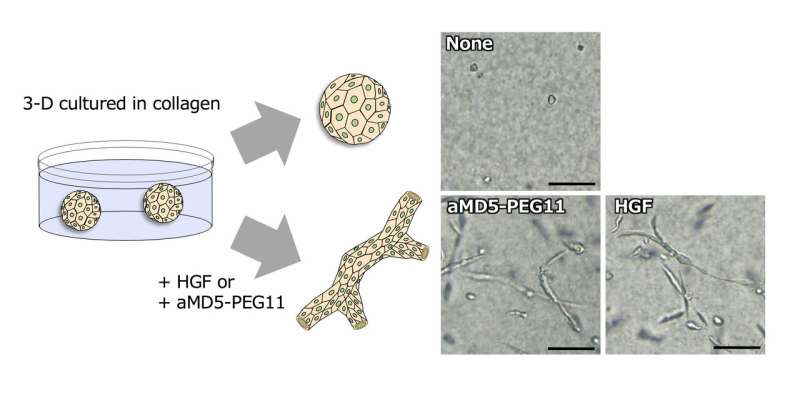
The activation of a set of genes is responsible for specific biological functions. When the genes activated by aMD-PEG11 and HGF were respectively analyzed during 3-D tubulogenesis in human kidney cells induced by aMD5-PEG11 and HGF, changes in the gene expression profiles were largely overlapped and comparable between HGF and aMD5-PEG11. Because 3-D tubulogenesis by HGF in kidney cells implicates construction/development and re-construction/regeneration of a functional kidney, the comparable ability between HGF and aMD5-PEG11 to induce gene expression profiles suggests that aMD5-PEG11 can facilitate regeneration of the kidney in a potential comparable to HGF (Fig. 2).
Several growth factors have been used as biological drugs manufactured by recombinant protein production. This study reported a synthetic artificial ligand that induced chemical and biological effects equivalent to a natural ligand. Not only are artificial ligands relatively cheaper to produce, but often their smaller size leads to greater permeability in the body. "Artificial MET receptor agonists such as macrocyclic peptides have the potential to be developed as novel biological drugs manufactured by chemical synthesis," the team concludes. Besides growth factors, this strategy can be applied to many kinds of ligands.
More information: Wenyu Miao et al, Cellular signaling and gene expression profiles evoked by a bivalent macrocyclic peptide that serves as an artificial MET receptor agonist, Scientific Reports (2018). DOI: 10.1038/s41598-018-34835-4
Journal information: Scientific Reports
Provided by Kanazawa University