How scientists analyze cell membranes
Exchange of material and information at the level of individual cells requires transport and signaling at the level of the plasma membrane enclosing the cell. Studying mechanisms at such tiny dimensions presents researchers with enormous challenges. Recently, researchers wanted to determine the function and distribution of cholesterol, an important component of the membrane. So far, cholesterol can only be labeled to a very limited extent with fluorescent dyes, which can be visualized under the microscope without damaging the membrane. Researchers at the University of Münster (Germany) have now developed a method to circumvent these difficulties. They synthesized a new compound with properties similar to those of cholesterol, but which can be labeled with dyes and visualized in living cells. There, the compound realistically mimics the behaviour of natural cholesterol.
"Our new approach offers enormous potential for imaging membrane dynamics in living cells," says Prof. Volker Gerke, one of the leaders of the study. The work is the result of an interdisciplinary study involving organic chemists, biochemists and biophysicists. The study appears in the current issue of the journal Cell Chemical Biology.
Cells in the body are enclosed in a kind of protective envelope, the plasma membrane, which separates the cell from its environment. Cells also contain internal membranes separating the individual components from each other and regulate the movement of substances between the different internal spaces. Cholesterol, a fatlike substance, is an important component of membranes ensuring that they work properly.
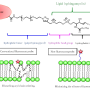
In order to generate substances that behave similarly to natural cholesterol, the organic chemists led by Prof. Frank Glorius first synthesized a series of chemical compounds. As a starting substance, they used natural cholesterol, which was transformed into a certain organic salt, an imidazolium salt. "We already knew from previous studies that these salts interact well with biomolecules and are therefore suitable for cellular experiments," says Frank Glorius, who also led the study.
In order to compare the biophysical properties of the newly synthesized compounds with those of the natural cholesterol, the researchers incorporated the substances to synthetic model membranes consisting of phospholipids (these phospholipids constitute the main component of membranes). Biochemists and biophysicists in the group of Prof. Dr. Hans-Joachim Galla measured how the new substances affected the phase transition temperature of model membranes, and how they changed the fluidity in the phospholipid layer at different temperatures. "After evaluating the data, we finally settled on three compounds that exhibited very similar properties to those of natural cholesterol," says Lena Rakers, a Ph.D. student of Organic Chemistry and one of the two first authors of the study.
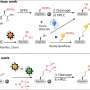
The researchers selected these compounds in order to examine them in living cellular membranes, thereby studying them in even more complex structures. For this purpose, they used cultures of human epithelial cells—HeLa cells—as well as cells from human blood vessels, HUVEC cells. Due to their structure, the newly synthesized substances fitted well into the cellular membranes. With the aid of surface mass spectrometry, the researchers measured the molecules in the membrane and could show that the compounds behaved in a very similar way to natural cholesterol in living cells, too.
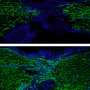
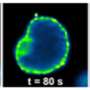
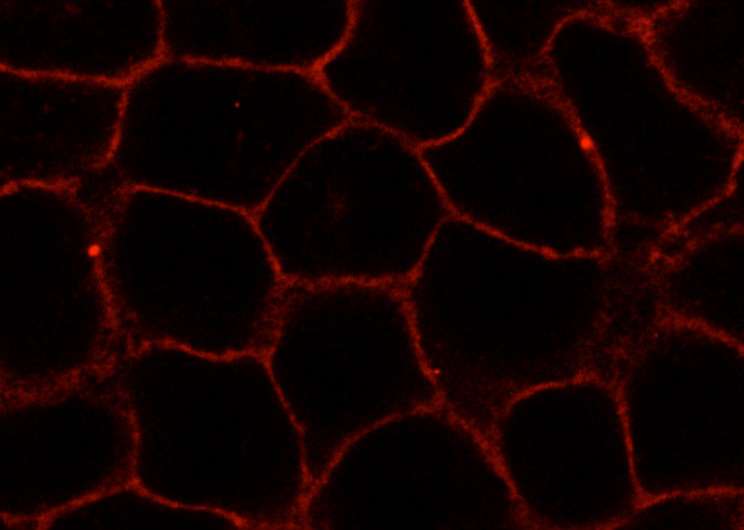
Because of its structure, one of the new substances could be labeled with fluorescent dyes. To this end, the researchers attached an azide group onto the substance. They then linked the dyes to this azide group using click chemistry—an effective method enabling molecular components to be joined on the basis of a few chemical reactions. Finally, the biochemists visualized the substance in living cells using high-resolution confocal microscopy. In this way, they were able to observe its distribution and dynamic changes. "These analyses also showed that the novel compound behaved analogously to cellular cholesterol," says David Grill, a Ph.D. student of Biochemistry and the other first author of the study. One great advantage of the new method is that during the entire process, the components and the properties of the cellular membrane remained undamaged.
In the future, the researchers want to continue developing their method and test the new substances in further cellular studies using microscopic imaging methods. One of their aims is to use click chemistry to attach fluorescent dyes and other molecules to the new compounds to introduce selective changes in the membrane.
More information: Novel addressable cholesterol analogs for live imaging of cellular membranes. Cell Chem Biol, 2018, DOI: 10.2016/j.chembiol.2018.04.015
Journal information: Cell Chemical Biology
Provided by University of Münster