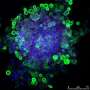
Cells grow more naturally in 'spaghetti'

The usual way of cultivating cells is to use a flat laboratory dish of glass. However, inside a human body, the cells do not grow on a flat surface, but rather in three dimensions. This has lead researchers at Lund University in Sweden to develop a porous "spaghetti" of tissue-friendly polymers with cavities in which the cells can develop in a more natural way.
"When cultivating brain cells in a flat laboratory dish, the different cell types form layers, with the nerve cells on top and the glial cells – a form of supporting tissue –underneath. This is not what it looks like in natural brain tissue, where the cells are much more mixed," says neuroscience researcher Ulrica Englund Johansson.
Many research groups around the world have therefore tried to develop three-dimensional structures in which cells can be cultivated in a more natural way. The Lund researchers have used a method called electrospinning.
"Electrospinning is actually an old technique, which has received a recent boost. It proved to be good way to produce small nanostructures for biological and medical purposes," explains biophysicist Fredrik Johansson, who works closely with Ulrica Englund Johansson's group.
The type of polymer used has been approved for medical purposes, and is used for e.g. sutures where the fibre eventually dissolves itself. Depending on the application, the three-dimensional structure can be shaped into different forms.
"You can let the fibres form a tangle with many cavities in which cells can grow, like a ball of boiled spaghetti. But if you, for example, want to get the neurite to grow in a certain direction, you can make the fibres form parallel lines – like straight, uncooked spaghetti," explains Fredrik Johansson by using a metaphor that is easy to understand.

The Lund researchers have achieved good results with their three-dimensional fibre structures.
"The three-dimensional shape seems to benefit the maturation of stem cells into glial cells and neurons. They also blend naturally together, develop long neurite outgrowths, and demonstrate functional electrical activity," says Ulrica Englund Johansson.
"They also express the proteins which are normally expressed in vivo. This indicates that the stem cells develop into the nerve cells they would have become in the brain."
If the new technique delivers what it promises, electrospinning will be able to provide new opportunities for both research and industry. With more natural cell cultures on which to conduct research, a number of biomedical research issues can be addressed in new ways.
New potential drugs can be tested more effectively on cell cultures that more closely resemble natural tissue. Cells which are to be transplanted – e.g. to the retina or to the brain – will probably also survive and develop better in a three-dimensional structure, even if they are later injected simply as cells in a solution.
The collaborating researchers, which also include biologist David O'Carroll, have recently published their results in three international journals: Nanomedicine, Journals of Biomaterials and Nano Biotechnology, and Molecular and Cellular Neuroscience. The first two articles describe their studies performed on human brain stem cells, while the third is about experiments using retinal cells.
More information: Albin Jakobsson et al. Three-dimensional functional human neuronal networks in uncompressed low-density electrospun fiber scaffolds, Nanomedicine: Nanotechnology, Biology and Medicine (2017). DOI: 10.1016/j.nano.2016.12.023
Journal information: Nanomedicine , Biomaterials , Molecular and Cellular Neuroscience
Provided by Lund University