New information about bacterial enzymes to help scientists develop more effective antibiotics, cancer drugs
Scientists studying the biosynthesis and production of microbial natural products now have a greater insight into the process thanks to research conducted at the U.S. Department of Energy's Argonne National Laboratory in collaboration with scientists from the Scripps Research Institute and Rice University.
Armed with this new information, researchers can use it to manipulate nature's biosynthetic machinery to produce more effective antibiotics and cancer-fighting drugs.
Streptomyces are Gram-positive bacteria that live in soil. These bacteria possess a complex metabolism and are known to naturally produce clinically useful compounds.
One large class of natural products, known as polyketides, includes many drugs such as erythromycin (antibacterial) and rapamycin (immunosuppressive), as well as promising drug leads such as migrastatin and oxazolomycin reported in the current study, which show important antibacterial, antitumor, and anti-human immunodeficiency virus activity.
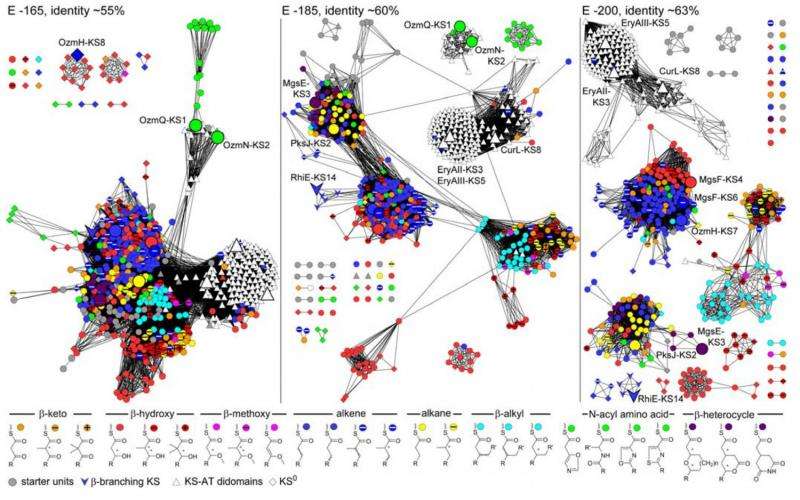
These antibiotics are synthesized by a set of enzymes that are orchestrated into assembly-line-like biosynthetic machinery. Researchers in this study focused on understanding the enzymes specificity, which is responsible for generating the vast chemical structural diversities known for migrastatin, oxazolomycin and other polyketides.
Andrzej Joachimiak works in the biosciences division at Argonne and was one of the authors of a recent paper published on the topic in The Proceedings of the National Academy of Sciences.
"If we understand the specificity of these processes, we will be able to engineer the enzymes to accept other chemical molecules, opening the door to new treatments for some of our most challenging diseases," said Joachimiak, director of the Department of Energy's Structural Biology Center, which is located at Argonne.
Antibiotics are made up of a set of multiple enzymes that perform consecutive actions.
Scientists seek to modify these molecules chemically to create new drugs with improved therapeutic properties.
"In order to do that we need to understand the specificity of this "enzymatic assembly line," Dr. Ben Shen of the Scripps Research Institute said. "We need to know which part we need to place, and do it in a rational and specific manner, to synthesize the designer compounds."
Manipulating enzymes that catalyze complex reactions that alter natural product structures to create diverse novel compounds with new biological activities is a key.
This work was done with the help of the Advanced Protein Characterization Facility, which has greatly aided medical and biomedical research by automating the production of protein and protein crystals - two key steps in solving the structure of proteins, understanding how they operate and ultimately helping to identify new and more effective drug treatments.
Proteins are long molecular chains that fold on themselves in complex ways with many of those folds serving as docking sites where other molecules - including those from pathogens - can attach.
In protein structure research, snippets of the DNA code for a protein are cloned. The clones are used to produce the proteins that are isolated and exposed to various chemical environments with the hope that one of them will cause the protein molecules to form a crystal.
This can take days, weeks or even months. But when it happens, the protein molecules align to form a repeating array. That repetitive configuration allows X-rays from the Advanced Photon Source, a DOE Office of Science User Facility located at Argonne, to analyze the three-dimensional structure of the molecules by means of their different signatures.
This helps Joachimiak and his team to solve age-old problems.
"This work would not be possible without the technology and equipment available here at Argonne," he said.
More information: Jeremy R. Lohman et al. Structural and evolutionary relationships of "AT-less" type I polyketide synthase ketosynthases, Proceedings of the National Academy of Sciences (2015). DOI: 10.1073/pnas.1515460112
Journal information: Proceedings of the National Academy of Sciences
Provided by Argonne National Laboratory