How the motility structure of the unicellular archaea is fixed to their surface
A research team led by the Freiburg microbiologist Prof. Dr. Sonja-Verena Albers has described the structure of the protein with which the motility structure is fixed to the cell wall of archaea – a type of unicellular life form. In addition, the researchers demonstrated that this protein is essential for the structure and functioning of the organ. The researchers published their findings in the current issue of the journal Structure.
Prokaryotes, unicellular life forms without a cell nucleus, are subdivided into bacteria and archaea. Much less research has been conducted on archaea, because no pathogenic forms have yet been described. The first isolated archaea came from habitats like hot sulfur springs, hot springs in the deep sea, or extremely saline lakes. Scientists thus long assumed that archaea can only grow in extreme environments like these. Now it is known that archaea, like bacteria, may be found in almost all habitats – in the intestinal flora and on the skin of humans, among other places. These discoveries provided a stimulus for new research.
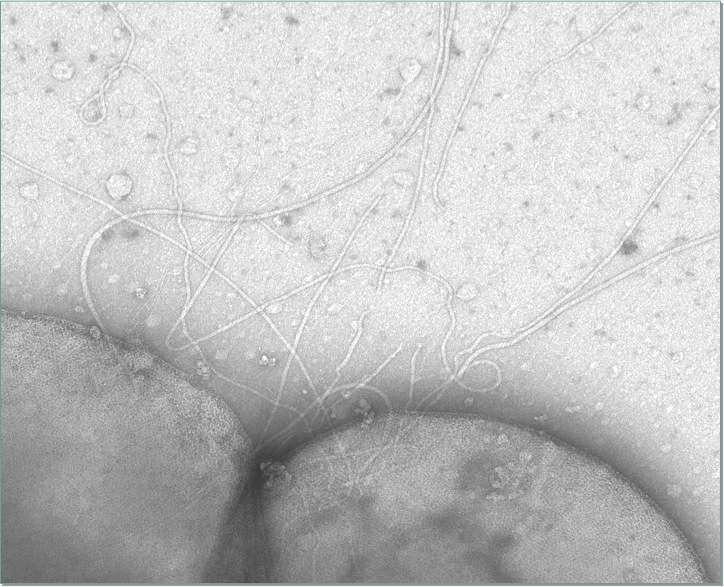
It is important for microorganisms to be able to move on their own: When their living conditions deteriorate, they are then capable of finding better ones on their own. The motility structure of bacteria, the flagellum, has been the object of detailed research for more than 30 years. It consists of up to 50 proteins assembled according to a fixed sequence of events. The result is a whip made of protein filaments that functions much like a propeller: A "motor" at the end fixed to the cell wall allows it to rotate, enabling the bacterium to swim.
Up until only a few years ago, scientists assumed that archaea also use flagella to move. However, the sequencing of the first archaeal genome revealed clear differences in the structures of the motility structures of bacteria and archaea. It was found that archaea use a structure called the archaellum to swim. In the model organism Sulfolobus acidocaldarius, it consists of a mere seven subunits but still achieves the same performance as the flagellum despite this simple structure. Only few structural studies of the subunits that make up the archaellum have been conducted to date. Two years ago Albers' research team discovered the structure of the motor protein FlaI and demonstrated that it forms the motor complex of the archaellum along with the proteins FlaX and FlaH. In their newly published article, the researchers describe the protein FlaF, which binds specifically to the model organism's only cell wall protein and fixes it firmly there. "It is important to learn more about these cell wall and surface structures, since the archaea can use them to interact with the environment – and thus also with human cells," says Albers.
More information: "FlaF Is a β-Sandwich Protein that Anchors the Archaellum in the Archaeal Cell Envelope by Binding the S-Layer Protein." Structure. dx.doi.org/10.1016/j.str.2015.03.001
Journal information: Structure
Provided by Albert Ludwigs University of Freiburg