This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New biocatalytic method eliminates cyanide in nitrile production
If the household cleaner emits a lemon-like odor, this may be due to a nitrile called citronellyl nitrile. These versatile chemical nitrile groups are also used in the manufacture of active pharmaceutical ingredients, superglue and chemical-resistant gloves. The prevalent production process used so far has required a chemical reaction of certain molecules with highly toxic cyanide.
Margit Winkler from the Institute of Molecular Biotechnology at TU Graz, together with Ludmila Martínková from the Institute of Microbiology at the Czech Academy of Sciences, has now developed a biocatalytic process that does not require cyanide and works at room temperature, therefore it requires less energy and produces less harmful waste.
The work is published in the Journal of Biotechnology.
Two enzyme-guided steps
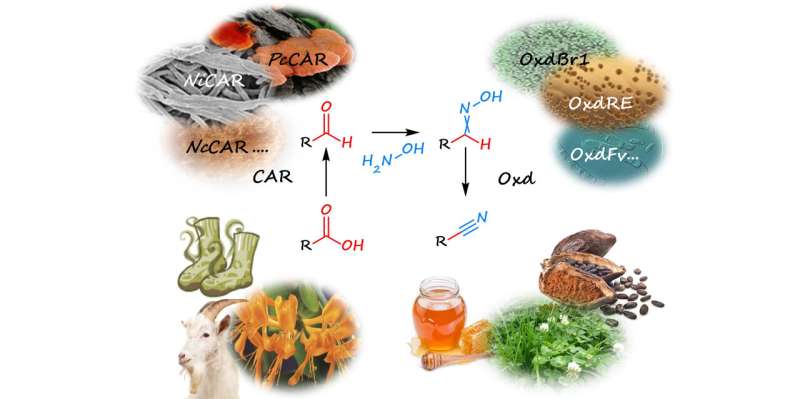
To replace the cyanide, the research group uses enzymes and combines three individual reactions into a cascade. The starting materials are carboxylic acids, such as fatty acids from plant oils or lignin, which is a by-product of wood processing.
In the first step, the carboxylic acid is converted to an aldehyde by adding sugar, oxygen and the enzyme carboxylate reductase contained in living bacterial cells. However, this aldehyde is highly reactive and therefore unstable, and in many cases also volatile.
To capture this substance, the aldehyde is allowed to react with hydroxylamine, which converts it into oxime, which is stable.
Oxime is a "watered" form of nitrile, which is why the enzyme aldoxime dehydratase is used in the final step. This enzyme is also confined in bacterial cells and is responsible for water abstraction from the oxime.
Once this has been done, the cascade has achieved its objective and the desired nitrile has been produced.
This method can now be used for the preparation of small nitrile quantities. This is already a promising technology for very potent fragrances, for example, of which only a small amount is needed to achieve an effect.
Optimization work is still required for the production of larger quantities of nitrile in order to be able to use the biocatalytic process at reasonable costs. Currently, highly diluted solutions are used in the first process step and a large reactor would be required to produce large quantities, which is not economical yet.
Winkler is therefore already conducting research on how to make the first step more efficient. Dehydration of the oxime alone, on the other hand, is extremely efficient and has already reached technical maturity for production today.
"I've been interested in cyanide chemistry ever since I wrote my master thesis, and as we know from Agatha Christie's novels, cyanide is highly toxic," says Winkler. "It was therefore important for us to find a way to avoid this hazardous substance in production.
"The fatty acids we use come from vegetable oils. So, we really do have a bio-based production method here. We would now like to improve it even further so that it can be used widely."
More information: Barbora Křístková et al, Immobilization of aldoxime dehydratases on metal affinity resins and use of the immobilized catalysts for the synthesis of nitriles important in fragrance industry, Journal of Biotechnology (2024). DOI: 10.1016/j.jbiotec.2024.02.005
Journal information: Journal of Biotechnology
Provided by Graz University of Technology