This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New modular flow platform for improved SuFEx click chemistry
Researchers at the Flow Chemistry group of the University of Amsterdam's Van 't Hoff Institute for Molecular Sciences have developed a modular flow chemistry platform for a safe and efficient execution of SuFEx (Sulfur(VI) Fluoride Exchange) click chemistry.
In a paper just published in Nature Synthesis, they describe how their platform generates the toxic gaseous reagent sulfuryl fluoride in a safe and controlled manner, and how it facilitates the subsequent fast and selective functionalization of small molecules, peptides and proteins for therapeutic purposes.
Click chemistry—the subject of the 2022 Nobel Prize in Chemistry—is a powerful and efficient method for rapidly connecting chemical fragments. A popular, highly versatile type of click chemistry involves the so-called SuFEx (Sulfur(VI) Fluoride Exchange) reaction. It results in the formation of covalent bonds under mild conditions and can greatly simplify the synthesis of therapeutically relevant small molecules, peptides, and proteins.
Straightforward, safe and scalable
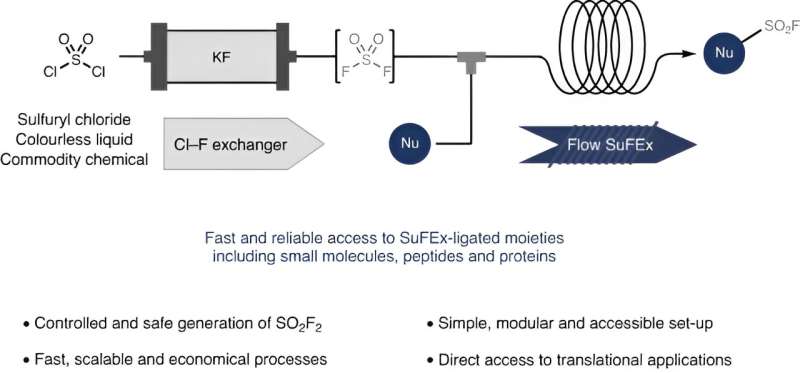
SuFEx chemistry involves attaching a -SO2F molecular moiety, after which the F atom can easily be replaced with a range of functional molecular groups of therapeutic value. An obvious reagent for introducing the -SO2F moiety is the gaseous component sulfuryl fluoride SO2F2, but since this is mildly toxic it requires elaborate synthetic procedures to avoid its direct use. In their Nature Synthesis paper, the Amsterdam chemists now present a modular system using microfluidic technology that renders the overall SuFEx procedure straightforward, safe and easily scalable.
The system consists of two interconnected flow reactors. The first generates sulfuryl fluoride in a controlled and dosed manner from cheap commodity chemicals sulfuryl chloride (SO2Cl2) and potassium fluoride (KF). In the second reactor, the generated gaseous sulfuryl fluoride is mixed with other reactants, ultimately yielding the desired SuFEx product. By immediately reacting away the sulfuryl fluoride, the modular system effectively eliminates the safety and practical concerns of the toxic reactant.
While exploring the performance of their system, the researchers were able to obtain a diverse range of SuFEx products with excellent yields in just two minutes of residence time. They attribute this short residence time to the intimate contact between gas and liquid phase in a flow system.
Full SuFEx-based click chemistry
The researchers report that a wide variety of natural products, drugs, and fluorescent tracers could be successfully reacted using the novel platform. They mention in particular that is now possible to use sulfuryl fluoride as a viable reagent for installing the -SO2F handle on a variety of phenol and amino functionalities. They expect their platform to facilitate the generation of large libraries of SuFEx compounds with minimal effort and time.
Capitalizing on the modular nature of their approach, the researchers developed a multistep system to perform full SuFEx-based click chemistry. They demonstrated excellent functional group tolerance and selectivity for a range of therapeutically relevant small molecules, peptides and proteins. For many cases, their system outperformed "traditional" experimental procedures reported in literature. Regarding direct protein modification, it turned out to be one of the fastest methods reported to date.
Overall, they consider their novel modular flow system an invaluable tool, facilitating remarkably swift and high-yielding SuFEx reactions between sulfuryl fluoride and an extensive array of structurally diverse substrates.
More information: Miguel Bernús et al, A modular flow platform for sulfur(VI) fluoride exchange ligation of small molecules, peptides and proteins, Nature Synthesis (2023). DOI: 10.1038/s44160-023-00441-0
Journal information: Nature Synthesis
Provided by University of Amsterdam