This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Scientists harness flower 'super power' to pave the way for new drug treatments
Scientists at the University of Bath have used nature as inspiration in developing a new tool that will help researchers develop new pharmaceutical treatments in a cleaner, greener, and less expensive way.
Drug treatments often work by binding to proteins involved in disease and blocking their activity, which either reduces symptoms or treats the disease.
Rather than using conventional small molecules as drugs, which are not well suited to blocking interactions between proteins, the pharmaceutical industry is now investigating the potential of making drugs using small proteins known as 'peptides,' which work in a similar way.
However, peptides and proteins often don't make very good drugs because their 3D structures can unravel, they are sensitive to high temperatures, and can be difficult to get inside the body's cells, where many exciting but challenging drug targets are found.
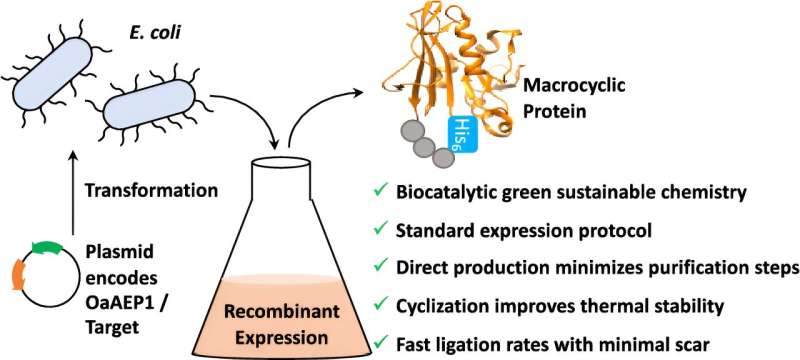
Now, scientists at the University of Bath have developed a way to get around this problem; normally, proteins and peptide strands have a start and an end—by joining these loose ends together, it is possible to create very rigid "cyclic" proteins and peptides which improves heat and chemical stability as well as making it easier to get them into cells.
They took an enzyme called OaAEP1 from Oldenlandia affinis, a small purple flower that grows in the tropics, and modified it before transferring it into bacterial cells. These bacterial cultures were grown to mass-produce a protein while simultaneously joining up the ends in a single step.
Plants can do this process naturally, but it is slow and low-yielding. Alternatively, cyclization can be done chemically by isolating the enzyme and then mixing multiple reagents in a test tube, but this requires multiple steps and uses toxic solvent chemicals.
Putting the entire process into a bacterial system increases the yield, uses more sustainable, biologically friendly reagents, and requires fewer steps. It is, therefore, much simpler and cheaper.
To demonstrate the approach, the scientists applied their bacterial OaAEP1 technology to a protein called DHFR, and found that joining together its head and tail ends made it more resistant to temperature changes while still retaining its normal function.
Professor Jody Mason, from the University of Bath's Department of Life Sciences, said, "Proteins and peptides are generally quite sensitive to heat, but cyclization makes them much more robust."
"The Oldenlandia plant naturally makes cyclic proteins as part of a defense mechanism to deter predators."
"So we've harnessed this flower superpower by modifying OaAEP1 and combining it with existing bacterial protein-producing technology to create a really powerful tool that will help the drug discovery industry."
Dr. Simon Tang, a Research Associate from the University of Bath's Department of Life Sciences, said, "Proteins and peptides are very promising as drug candidates, but a significant bottleneck for the development of new therapeutic treatments is producing enough of the stuff to reach patients without incurring an astronomical cost."
"Our new process lets the bacteria do all the work—the result is it's also cleaner and greener, and because it has fewer steps, it is a lot simpler to do."
"We're really excited about the potential applications of this, not only for the pharmaceutical industry but other industries such as the food industry, detergent industry, in biotechnology, and in bioenergy production."
The researchers have filed a patent for the technique and published their work in the journal JACS Au.
More information: T. M. Simon Tang et al, Intracellular Application of an Asparaginyl Endopeptidase for Producing Recombinant Head-to-Tail Cyclic Proteins, JACS Au (2023). DOI: 10.1021/jacsau.3c00591
Provided by University of Bath