This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Discovery of histidine methylation as a new post-translational modification of histone proteins
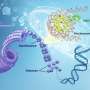
In eukaryotes, genomic DNA, which is a very long double helix containing all the genetic information, wraps around a globular protein called a histone and folds it many times before being contained within the nucleus.
Various post-translational modifications (for example, addition of chemical groups) occur on histones. Among them, the methylation of residues of lysine, which is one of the amino acids that make up histones, regulates the folding of genomic DNA and acts as a switch to turn gene transcription on and off.
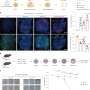
A research group has discovered the methylation of histidine residues as a new post-translational modification of histones using a unique method to distinguish precisely the presence and mode of methylation of proteins. Their work has been published in the Journal of Biological Chemistry.
Histones form an octamer, which contains two copies each of the four core histone proteins H2A, H2B, H3, and H4. Among these, histidine methylation was found to occur at the 82nd histidine of histone H2A and at the 39th histidine of histone H3. Additionally, an examination of all the methylation states of histone H3 showed that most methylation modifications were concentrated on lysine residues, suggesting that methylation of histidine residues in histones occurs only in few histones in a specific gene region.
Histones contain many lysine residues, which undergo various post-translational modifications such as methylation and acetylation. The combination pattern is called the histone code, which is regarded as a code that directs transcriptional regulation. The discovery of the methylation of histidine residues is expected to be a new step toward deciphering the histone code.
More information: Takahiro Hayashi et al, Histidine Nτ-methylation identified as a new posttranslational modification in histone H2A at His-82 and H3 at His-39, Journal of Biological Chemistry (2023). DOI: 10.1016/j.jbc.2023.105131
Provided by University of Tsukuba