Investigating better biotransformations

Research in the International Journal of Bioinformatics Research and Applications has investigated the chemistry and behavior of a useful natural product made by the Madagascar periwinkle (Catharanthus roseus Bunge). The work could expand the repertoire of a growing area of chemical science—biotransformation—wherein nature's molecular machinery is used to build and alter novel compounds in the laboratory.
Many natural products, by definition chemical compounds made by living organisms, have physiological activity and have been isolated from their source and researched and developed into pharmaceutical products. Indeed, approximately two in every five prescription drugs have an origin in natural products. Commonly, however, the active chemical in a living organism is modified for a particular purpose or drug profile with different, more targeted activity in a disease, and fewer, or less harmful, side effects, for instance. In addition, modifying a natural product is often a prerequisite to making a new pharmaceutical sufficiently different that a successful patent application can be made and a drug brought to market profitably.
In the last few decades, chemists have found ways to use enzymes to modify natural products and in turn, they have found ways to modify enzymes to make them work differently and allow them to process natural products and other molecules in different ways to generate unprecedented molecular diversity. Any one of these huge numbers of new molecules could have physiological activity that might be useful in treating particular diseases and disorders.
Piotr Szymczyk, Grażyna Szymańska, Małgorzata Majewska, Izabela Weremczuk-Jeżyna, Michał Kołodziejczyk, Kamila Czarnecka, Paweł Szymański, and Ewa Kochan of the Medical University of Łódź, in Łódź, Poland, have investigated part of nature's molecular machinery, an enzyme known as C. roseus strictosidine β-D-glucosidase. Enzymes are proteins that act on small molecules, their substrates, and convert that substrate into another molecule used by the living organism. The team reports the structure of this enzyme from the periwinkle with a focus on the pocket in its molecular structure that binds to the substrate, the enzyme's active site.
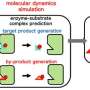
The team built a computer model of the periwinkle enzyme using Discovery Studio 4.1 software and a template for the enzyme based on another known enzyme from a β-glucosidase found in rice, which they modified to match the known details for the periwinkle enzyme. They could then use a second computer program—an algorithm called CDOCKER—to see how different chemical substrates would interact with the active site of the model periwinkle enzyme. They tested the natural substrate a molecule known as strictosidine and a second chemical D-glucono 1,5-lactone. This latter molecule is known to bind to the enzyme and inhibit its activity. The docking process in which substrate is put into the active site, like a key into a lock, then allowed the team to refine the structure of the periwinkle enzyme to make the fine details of the model closer to those seen in nature. To do this molecular dynamics software was used.
Ultimately, the work extends what was previously known about the periwinkle enzyme and could allow scientists to modify it in such a way to act on other substrates. Before that though, given that the natural product strictosidine itself is a useful starting material for a wide range of different molecules, the work opens up new avenues for working with this natural product.
More information: Ewa Kochan et al, Homology modeling and docking studies of strictosidine beta-D-glucosidase from Madagascar periwinkle (Catharanthus roseus Bunge), International Journal of Bioinformatics Research and Applications (2020). DOI: 10.1504/IJBRA.2022.10033607
Provided by Inderscience