Induced pluripotent bovine stem cells overcome decades-long challenges for cultivated meat

Stem cells have been used for years as therapeutics for human health, but new research shows they may offer the promise of making the global food supply more sustainable and reliable through the development of cultivated meat.
One type of stem cell that's of particular interest is known as induced pluripotent stem cells (iPS cells). These cells are derived from adult cells and can be reprogrammed to act as embryonic stem cells. These cells are pluripotent, which function as the building blocks that gradually give rise to every kind of organ and tissue cells.
That could be good news for the growing lab-grown meat industry, which seeks ways to bring meat to consumers without the environmental drawbacks of large-scale industrial farming. To date, however, the industry has been unable to generate enough high-quality livestock pluripotent stem cells to make this a legitimate commercial option. While scientists have successfully created iPS cells for human and mouse models, there has been a near standstill in the development of these pluripotent cells for cattle.
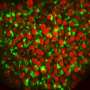
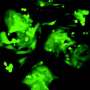
That may be changing: Associate Professor Young Tang and Professor and Interim Department Head Xiuchun (Cindy) Tian in the College of Agriculture, Health and Natural Resources recently demonstrated the first successful iPS cells for bovine.
The duo published their findings in International Journal of Molecular Sciences. They have filed a provisional patent on this technology through UConn Technology Commercialization Services.
Bovine pluripotent stem cells could have applications for lab-grown beef, as they could allow scientists to grow entire cuts of meat from muscle stem cells differentiated from a single iPS cell.
These cells could also be used for the establishment of in-vitro breeding technology, which theoretically could create 100 generations of cows in 25 years. Traditional breeding techniques can only produce 10 generations in the same period.
These cells would also allow great advances in genetic engineering and developing disease-resistant animals, according to the UConn research team.
"This will be very significant for our future, because we want to build up sustainable agriculture production and accommodate the increased need for the global population," Tang says
Silencing noisy genes
Tang and Tian's team used a novel combination of decades of stem cell research to finally overcome the barriers for bovine iPS cell development.
Previously, bovine stem cells had two major issues: they couldn't silence the genes used to reprogram them, and they couldn't renew themselves for the long term.
While a prerequisite of pluripotency is the capacity of the iPS cells to actively silence the transgenes that induce them, incompletely reprogrammed cells will die or differentiate within a couple of replication cycles when these genes are turned off manually.
Tang and Tian's cells overcome both issues by silencing a methylation event that was previously identified as a barrier for embryo development in mice and humans.
The researchers previously developed a method for creating iPS cells in other animal models. This method combined two widely used sets of proteins: Yamanaka factors (Oct4, Sox2, Klf4, and c-Myc) and Thompson factors (Oct4, Sox2, Lin28, and NANOG).
Traditionally, scientists use only one set of these reprogramming factors. But Tang and Tian combined them, using a total of six factors: Oct4, Sox2, Klf4, c-Myc, Lin28, and NANOG.
They also used two inhibitors to shut down signaling pathways that interfered with the reprogramming process.
These changes allowed the researchers to make human cell reprograming 100 times more efficient than using Yamanaka factors alone.
The next step was to shut down a methylation event in bovine chromatin known as Histone 3 Lysine 9 Trimethylation (H3K9me3). They found that overexpressing an enzyme known as histone lysine demethylase KDM4A effectively shut this methylation pathway down.
"Our hypothesis was that this H3K9 trimethylation represented a reprogramming barrier for the bovine iPS cell generation," Tang says, "Therefore, we combined our previous robust reprogramming system that we worked out using human cells, with the co-expression of KDM4A and we found that we could generate the bovine iPS cells."
What's next for iPS cells?
The next step for this technology will be testing how pluripotent they are. To determine this, the researchers will inject the cells into an embryo and analyze which tissues and organs the iPS cells reach.
The more pluripotent, the more tissue types the cells will incorporate into. Less pluripotent iPS cells may show little incorporation or be mainly confined to whatever tissue type they came from originally, a theory called epigenetic memory.
"They remember where they come from," Tian says. "And then they end up in that tissue."
The ultimate test of pluripotency is if the iPS cells can incorporate into the animal's reproductive system—either sperm or eggs—and pass their genes onto a new generation.
This research could apply to other animals like sheep and goats, or even near-threatened species of bovine, like bison or buffalo.
More information: Yue Su et al, Establishment of Bovine-Induced Pluripotent Stem Cells, International Journal of Molecular Sciences (2021). DOI: 10.3390/ijms221910489
Provided by University of Connecticut