Mystery of the nanobubbles solved

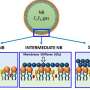
Water containing suspended nanobubbles is utilized in many biological and medical applications. For example, nanobubbles increase the efficiency of drug and gene delivery. Water containing oxygen nanobubbles is used in wastewater treatment, to promote the growth of plants and animals, and as a cure for diseases caused by anaerobic bacteria. The observed lifetime of suspended nanobubbles is up to several weeks. However, according to the classical theory of bubble stability, a nanosized bubble should dissolve within microseconds. This controversy between experiments and theory is known as the nanobubble paradox.
Now, researchers of VTT Technical Research Centre of Finland, Tapio Vehmas and Lasse Makkonen, have solved this paradox. According to their thermodynamic analysis, nanoscale bubbles indeed dissolve rapidly, but when the bubble is sufficiently small already upon its formation, the dissolving process does not begin. This is because when a very small bubble shrinks, the energy that is required to create the supersaturation necessary to transfer gas from the bubble boundary to the liquid, is more than the corresponding reduction in the surface energy of the bubble. According to Vehmas and Makkonen, the bubble diameter, below which dissolving does not begin in saturated water at room temperature, is 180 nanometers.
The study by Vehmas and Makkonen clarifies why nanobubbles can exist, and what is the basis of producing them. This will assist in developing more efficient nanobubble generators. The new theory can be applied also to nanobubbles attached to surfaces. Surface nanobubbles are utilized, for example, in cleaning, and in reducing friction of objects moving in water.
The study "Metastable nanobubbles" by Makkonen and Tikanmäki was published in American Chemical Society´s journal ACS Omega.
More information: Tapio Vehmas et al. Metastable Nanobubbles, ACS Omega (2021). DOI: 10.1021/acsomega.0c05384
Journal information: ACS Omega
Provided by VTT Technical Research Centre of Finland