Researchers realize homogenization of surface active sites of heterogeneous catalyst

Recently, a team led by Professor Wu Changzheng from School of Chemistry and Materials Science from University of Science and Technology of China (USTC) in cooperation with a team led by Prof. Wu Hengan from School of Engineering Science, realized the homogenization of surface active sites of heterogeneous catalyst by dissolving the electrocatalytic active metal in molten gallium. The related results have been published in Nature Catalysis on March 11th.
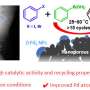
Due to the existence of various defects and crystal faces, the active components on the surface of heterogeneous catalysts are often in different local coordination environments. This leads to the coexistence of multiple active sites and the decrease of product selectivity.
The homogeneous catalyst has obvious advantages in product selectivity and and in a reaction mechanism research model. Therefore, it a key challenge will be to achieve the homogenization of heterogeneous catalysts.
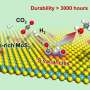
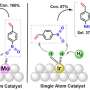
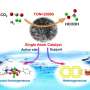
Faced with this challenge, Prof. Wu's group realized the homogenization of Sn and in heterogeneous catalysts by choosing molten metal Ga the near room temperature as the substrate of Sn and in with electrocatalytic activity for carbon dioxide reduction.
Compared with solid Ga, the active sites loaded in liquid Ga have significantly enhanced reducibility. The selectivity of formic acid is increased from about 30% in solid Ga to more than 95% in liquid Ga, and the side reaction of hydrogen evolution is obviously inhibited.
In addition, due to its good redox reversibility and fluidity of surface components, the liquid alloy exhibits self repairing properties and significantly improves its catalytic stability.
This work explored the feasibility of using liquid materials as the substrate of heterogeneous catalysts, which brings new insight for the synthesis of heterogeneous catalysts.
More information: Hongfei Liu et al, Solid–liquid phase transition induced electrocatalytic switching from hydrogen evolution to highly selective CO2 reduction, Nature Catalysis (2021). DOI: 10.1038/s41929-021-00576-3
Journal information: Nature Catalysis
Provided by University of Science and Technology of China