Why developing nerve cells can take a wrong turn
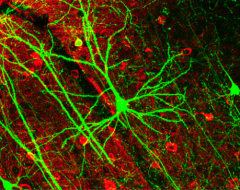
A group of scientists from CECAD has found a mechanism by which neurodevelopmental diseases concerning neurons can be explained. The loss of a certain enzyme, UBE2K, impedes the differentiation of stem cells by silencing the expression of genes important for neuronal differentiation and, therefore, the development and generation of neurons. More specifically, UBE2K regulates the levels and activation of histones, key proteins that pack and organize DNA, regulating the expression of genes. Being part of the epigenetic landscape of the cell, the changes made to the histones are reversible and could provide a chance for future treatments for neurodevelopmental diseases. The study is published in the current issue of Communications Biology.
Embryonic stem cells (ESCs) can replicate indefinitely while retaining their potential to differentiate into all other types of cells. Thus, nerve cells (neurons), muscle cells and all the other cells of the body are produced in a developing organism. Errors during this process can lead to congenital diseases. Degrading damaged proteins within the cell is an important factor in this process. Thus, the scientists studied the interaction between the proteasome, the main protagonist in terminating proteins, and the epigenetic landscape. Epigenetic landscapes are the heritable changes to an organism which are not determined by DNA but through changes to the chromatin, which can organize and silence DNA into tighter packages.
Azra Fatima from CECAD studied the interactions of the histones in immortal human embryonic stem cells (hESCs) which have a unique chromatin architecture and especially low levels of a certain histone called H3, which has undergone a chemical addition in the form of three methylgroups (H3K9me3).
Histones are proteins which are part of the chromatin in cell nuclei. They build up spools around which the DNA winds, shortening it by a ratio of 1:10 millions. They are also responsible for regulating the gene expression by which genes produce proteins in the organism. In addition, they play an important role in the process of cellular differentiation in which a cell, for example an embryonic stem cell, changes into another type of cell with a higher degree specialization.
They found that embryonic stem cells exhibit high expression of UBE2K (Ubiquitin-conjugating enzyme E2 K), a ubiquitin-conjugating enzyme. These enzymes are known for being important in the process of degradation of proteins. The loss of the enzyme in embryonic stem cells caused an increase in the levels of H3K9 trimethyltransferase SETDB1, resulting in higher trimethylation of H3K9, leading in turn to a repression of neurogenic genes during the differentiation of the stem cells. As a result, the loss of UBE2K impaired the ability of the stem cells to differentiate into neural progenitors a type of precursor cell that generate neurons and other cells of the nervous system.
Besides H3K9 trimethylation, the scientists found that UBE2K binds histone H3 to induce its polyubiquitination and degradation by the 26S proteasome. Notably, ubc-20, the worm orthologue of UBE2K, also regulates both histone H3 levels and H3K9 trimethylation in the germ cells of the model organism C. elegans. "Our results indicate that UBE2K crosses evolutionary boundaries to promote histone H3 degradation and reduce H3K9me3 repressive marks in immortal cells like human embryonic stem cells and germline cells," says Fatima.
"We found a link between the ubiquitin-proteasome system and epigenetic regulation in immortal stem cells," Fatima concluded. "It would be also interesting to see if UBE2K regulates the epigenetic state in other cell types like cancer cells." David Vilchez, the corresponding author of the manuscript, added: "We believe that our findings can have important implications to understand the development of the human brain."
By precisely regulating the levels of UBE2K it would be possible to determine the cell type specific epigenetic landscapes. Different diseases like Huntington's disease are associated with alterations in epigenetic marks. Since the epigenetic marks are reversible, it will be interesting to study if the epigenetic status of pluripotent stem cells from patients can be modulated by controlling the proteasome system and UBE2K. In order to correct the disease phenotype, novel strategies could be designed to correct epigenetic alterations in early developmental stages and thus provide a potential treatment for diseases.
More information: Azra Fatima et al. The ubiquitin-conjugating enzyme UBE2K determines neurogenic potential through histone H3 in human embryonic stem cells, Communications Biology (2020). DOI: 10.1038/s42003-020-0984-3
Journal information: Communications Biology
Provided by University of Cologne