Physicists determine the barely-measurable property entropy for the first time in complex plasmas

Since the end of the 19th century, physicists have known that the transfer of energy from one body to another is associated with entropy. It quickly became clear that this quantity is of fundamental importance, and so began its triumphant rise as a useful theoretical quantity in physics, chemistry and engineering. However, it is often very difficult to measure. Professor Dietmar Block and Frank Wieben of Kiel University (CAU) have now succeeded in measuring entropy in complex plasmas, as they reported recently in the renowned scientific journal Physical Review Letters. In a system of charged microparticles within this ionized gas, the researchers were able to measure all positions and velocities of the particles simultaneously. In this way, they were able to determine the entropy, as it was already described theoretically by the physicist Ludwig Boltzmann around 1880.
Surprising thermodynamic equilibrium in plasma
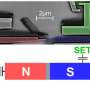
"With our experiments, we were able to prove that in the important model system of complex plasma, the thermodynamic fundamentals are fulfilled. What is surprising is that this applies to microparticles in a plasma, which is far away from thermodynamic equilibrium," explains Ph.D. student Frank Wieben. In his experiments, he is able to adjust the thermal motion of the microparticles by means of a laser beam. Using video microscopy, he can observe the dynamic behaviour of the particles in real time, and determine the entropy from the information collected.
"We thus lay the fundation for future fundamental studies on thermodynamics in strongly coupled systems. Theseare applicableto other systems as well," says Professor Dietmar Block from the Institute of Experimental and Applied Physics at the CAU. The origin for this success is largely attributable to the results and diagnostic techniques.
Explaining entropy with a water experiment
An everyday experiment illustrates entropy: If you pour a container of hot water into a container of cold water, the mixture is cooler than the hot water, and warmer than the cold water. However, you cannot undo this process—it is irreversible: Water at medium temperature cannot be split into a container of hot water and a container of cold water.
The reason for the irreversibility of this process is entropy. The second law of thermodynamics states that the entropy in a closed system never decreases over time. Therefore, the mixing of hot and cold water must increase the entropy. Alternatively, entropy can also be associated with the degree of disorder or randomness. In highly simplified terms, you could say that systems do not change into a more orderly state by themselves. Someone has to create order, but disorder can arise by itself.
More information: Frank Wieben et al, Entropy Measurement in Strongly Coupled Complex Plasmas, Physical Review Letters (2019). DOI: 10.1103/PhysRevLett.123.225001
Journal information: Physical Review Letters
Provided by Kiel University