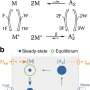
Theorem explains why quantities such as heat and power can fluctuate in microscopic system

The second law of thermodynamics states that the total entropy of an isolated system always tends to increase over time until it reaches a maximum. In other words, disorganization increases without outside intervention. Electrical equipment inevitably heats up as part of the energy is dissipated in the form of heat instead of being used for mechanical work, and objects deteriorate over time but do not spontaneously regenerate.
However, this intuitive nature of entropy does not necessarily apply to the microscopic world. Physicists have therefore reinterpreted the second law by giving it a statistical twist: Entropy indeed increases, but there is a non-null probability that it may sometimes decrease.
For example, instead of heat flowing from a hot body to a cold one, as usual, it may flow from a cold body to a hot one in certain situations. Fluctuation theorems (FTs) quantified this probability with precision, and the issue has practical interest in regard to the operation of nanoscale machines. FTs were proposed for the first time in an article published in 1993 in Physical Review Letters. The article was written by Australian scientists Denis Evans and Gary Morriss and Dutch scientist Ezechiel Cohen. They tested one of these theorems using computer simulations.
An article published recently in the same journal shows that one consequence of FTs is thermodynamic uncertainty relations, which involve fluctuations in the values of thermodynamic quantities such as heat, work and power. The title of the new article is "Thermodynamic uncertainty relations from exchange fluctuation theorems."
The first author was André Timpanaro, a professor at the Federal University of the ABC (UFABC), São Paulo State, Brazil. The principal investigator for the study was Gabriel Landi, a professor at the University of São Paulo's Physics Institute (IF-USP). Giacomo Guarnieri and John Goold, affiliated with Trinity College Dublin's Physics Department (Ireland), also participated.
Uncertainty relations
"The physical origins of thermodynamic uncertainty relations were obscure until now. Our study shows they can be derived from FTs," Landi said.
"When we began studying thermodynamics, we had to deal with such quantities as heat, work and power, to which we always assigned fixed values. We never imagined they could fluctuate, but they do. In the microscopic world, these fluctuations are relevant. They may influence the operations of a nanoscale machine, for example. Thermodynamic uncertainty relations establish a floor for these fluctuations, linking them to other quantities such as system size."
Thermodynamic uncertainty relations were discovered in 2015 by a group of researchers led by Udo Seifert at Stuttgart University in Germany. André Cardoso Barato, a former student at IF-USP and currently a professor at the University of Houston (U.S.), participated in the discovery.
The mathematical structure of these relations resembles that of Heisenberg's uncertainty principle, but they have nothing to do with quantum physics; they are purely thermodynamic. "The nature of thermodynamic uncertainty relations has never been very clear," Landi said. "Our main contribution was to show that they derive from FTs. We believe that FTs describe the second law of thermodynamics more generally and that thermodynamic uncertainty relations are a consequence of FTs."
According to Landi, this generalization of the second law of thermodynamics handles thermodynamic quantities as entities that can fluctuate, though not arbitrarily, since they must obey certain symmetries. "There are several fluctuation theorems," he said. "We found a special class of FTs and focused on them as cases of mathematical symmetry. In this manner, we transformed our problem into a mathematical problem. Our main result was a theorem of probability theory."
More information: André M. Timpanaro et al, Thermodynamic Uncertainty Relations from Exchange Fluctuation Theorems, Physical Review Letters (2019). DOI: 10.1103/PhysRevLett.123.090604
Journal information: Physical Review Letters
Provided by FAPESP