Engineered bacterial biofilms immobilizing nanoparticles enable diverse catalytic applications
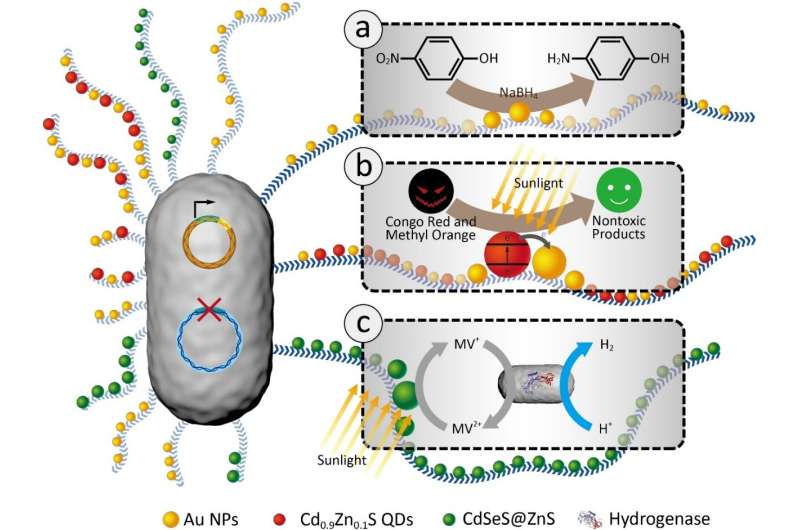
Immobilization is considered a feasible strategy for addressing toxicity and nanomaterial pollution confronted by nano-catalysts in practical applications. A research team from ShanghaiTech University harvested genetically engineered Escherichia coli biofilms as living substrates to immobilize nanoscale catalysts. The biofilm matrix provides a benign and robust interface between nano-catalysts and living cells, upon which three tunable and recyclable catalytic reaction systems has been demonstrated.
Nano-scale objects (1 -100 nm) are desirable nano-catalysts featured with more catalytic active sites due to higher surface-area-to-volume ratios. The nano-scale nature brings several attendant challenges such as leakage of nano-catalysts to ambient environment and difficulties in reusing nanocatalysts over repeated reaction cycles. A major strategy for addressing these challenges has been the immobilization of nano-objects on various substrates via a variety of technological approaches. However, inorganic and bio-derived or bio-inspired substrates obviously lack "biology-only" attributes like self-regeneration, cellular-growth-based scalability, and the ability of cells to biosynthesize complex enzymes, substrates, co-enzymes, or other required reagents or reaction components in situ. Moreover, studies that have immobilized nano-objects directly on cell surfaces have reported damage to cells.
The Zhong group from the Materials and Physical Biology Division, at ShanghaiTech University has made a major conceptual advance in developing a new abiotic/biotic interface towards the integration and immobilization of nanoscale objects with living cells for catalysis. Very briefly, they successfully showed how engineered amyloid monomers expressed, secreted and assembled in the extracellular matrix of living Escherichia coli (E. coli) biofilms can be harnessed to anchor functional nano-scale catalysts to make highly efficient, scalable, tunable, and reusable living catalyst systems. In their proof-of-concept studies, they have demonstrated three simple catalytic systems, including biofilm-anchored gold nanoparticles to degrade the pollutant p-nitrophenol, biofilm-anchored hybrid Cd0.9Zn0.1S quantum dots (QDs) and gold nanoparticles to efficiently degrade organic dyes, and biofilm-anchored CdSeS@ZnS QDs in a dual bacterial strain semi-artificial photosynthesis system for hydrogen production. As revealed in their studies, the extracellular matrix in biofilms indeed provides an ideal milieu for interfacing and anchoring nano-objects for direct catalysis and for their integration with the metabolism of living cells: even after multiple rounds of reactions, nano-catalysts were still robustly anchored to biofilms and the E. coli cells were still alive for easy regeneration. Importantly, such an approach would open up the extremely powerful and unique attributes of living systems.
There is a large diversity of bacterial biofilms with different functionalities in nature, and their study thus lays the conceptual foundation for coupling the uniquely dynamic properties and capacities of these living materials with the highly reactive nanoparticles to innovatively solve challenges in bioremediation, bioconversion, and energy. Their research will spur further research for creating more efficient and industrially important reaction systems by building and integrating more intricate biofilms/inorganic hybrid catalytic systems.
More information: Xinyu Wang et al, Immobilization of functional nano-objects in living engineered bacterial biofilms for catalytic applications, National Science Review (2019). DOI: 10.1093/nsr/nwz104
Provided by Science China Press