The perfect atomic-scale sieve
Graphene is perfectly selective to protons and blocks even smallest ions like chlorine, University of Manchester research shows. This result will be important for the development of graphene-based membranes for applications ranging from fuel cells to desalination.
Writing in Nature Communications, a team led by Dr. Marcelo Lozada-Hidalgo and Professor Andre Geim shows that 2-D materials like graphene and hexagonal boron nitride, also known as 'white graphene," are impenetrable to all ions. Only protons can transport through these crystals, which allows membranes with perfect proton selectivity.
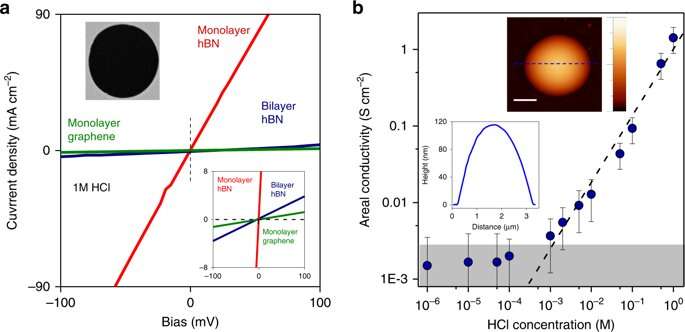
The researchers had previously found that protons easily permeate through graphene's crystal lattice. However, it remained unknown whether other small ions could pierce the dense crystal lattice in these materials. Now the researchers have found only protons can.
Besides its relevance for the development of 2-D crystal membranes, the work further supports the previous conclusion that holes in the crystal lattice are not necessary for proton transport through 2-D crystals. The 2-D crystal itself is highly permeable for protons.
Lucas Mogg, a Ph.D. student on the project and the first author of the paper said: "In our experiments, the 2-D crystal membranes separate reservoirs that contain both protons and chlorine ions. These reservoirs are practically infinite compared to our 2-D crystal membrane's size. We were very surprised to see that a one-atom-thick barrier was enough to stop all chlorine ions from crossing. Even thick polymer membranes especially designed to separate ions sometimes fail to achieve such perfect selectivity."
These findings are relevant for theory developments in the field of 2-D ionic conductors, adds Dr. Lozada-Hidalgo. "Our results conclusively show that proton transport through the two-dimensional crystals occurs through their bulk and does not require atomic-scale defects. This is an important development in our understanding of the interaction between ions and atomically-thin crystals with implications that extend well beyond the present study."
The results are also thought to be important in the development of a wide range of applications that use graphene as membrane materials. "Our results have implications for technologies that use graphene as a membrane material. The fast proton permeation of protons through the pristine 2-D crystal bulk is typically not taken into consideration. However, it could be important for designing and optimizing these membranes, especially when operating in acidic conditions," explains Marcelo Lozada-Hidalgo.
The researchers are enthusiastic about the prospects opened by this work. They believe that many more crystals could be studied using a similar approach. Most 2-D crystals remain unexplored from this perspective. The researchers think more unexpected phenomena and new applications could be found in these new materials.
More information: L. Mogg et al. Perfect proton selectivity in ion transport through two-dimensional crystals, Nature Communications (2019). DOI: 10.1038/s41467-019-12314-2
Journal information: Nature Communications
Provided by University of Manchester