February 26, 2016 feature
Tiny filters, big news: Novel process uses graphene and boron nitride monolayers to separate hydrogen ion isotopes
(Phys.org)—Conventional membranes used for sieving atomic and molecular species cannot scale to the subatomic level, making them unable to separate hydrogen isotope ions (protons, deuterons and tritons). At the same time, there are no current methods of directly separating these isotopes, and current approaches are extremely energy-intensive and therefore expensive – sometimes prohibitively so. Recently, however, scientists at the University of Manchester (UK) demonstrated a novel, scalable and highly competitive approach that uses monolayers of graphene and boron nitride as extremely fine sieves to separate hydrogen isotopes. Moreover, in addition to the new approach's simple and robust sieving mechanism, it offers straightforward setups and the need for only water as input without requiring additional chemical compounds.
Dr. Marcelo Lozada-Hidalgo and Phys.org discussed the paper that he and his colleagues, led by Regius Professor & Royal Society Research Professor Sir Andre Geim, published in Science. "In our previous investigation1 of whether deuterons permeate through 2D crystals differently from protons, there was certainly a lot of hard work involved," Lozada-Hidalgo tells Phys.org. "We had to fabricate a large number of devices, and reported on around 50, for this project in order to collect robust statistics – but the main challenge was explaining the results." The scientists were expecting deuterons to permeate only slightly slower than protons (perhaps a factor of 1.5 or so, but certainly not a factor of 10, Lozada-Hidalgo says). "On top of that, the existing theory did not predict any difference at all! We figured it out at the end but it was challenging because the field is highly interdisciplinary, being at the intersection of physics, chemistry and materials science, and is also very new – only two years old – so there's still a lot to figure out. Then again, that makes it all the more exciting."
One counterintuitive result, Lozada-Hidalgo adds, was finding the same isotope effect for all crystals – a differential factor of ten in permeation between protons and deuterons. We finally managed to understand all of it – but it certainly was puzzling for awhile."
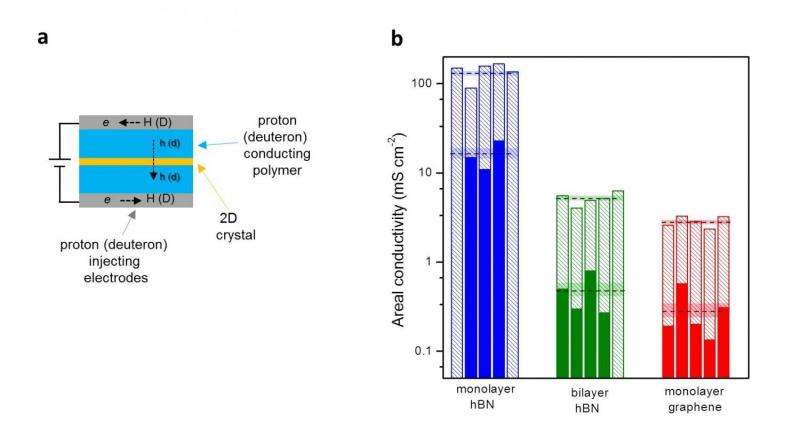
![Isotope effect measured by mass spectrometry. (a) Mass spectrometry set up. Pt decorated graphene is used to separate two chambers: one containing a proton-deuteron electrolyte mixture and another evacuated and facing a mass spectrometer. The graphene membrane is biases against the electrolyte mixture and three possible gases flow (HD, D2 or H2). (b) Fraction of protium atoms at the output for different proton inputs. The solid red curve shows [H] the theoretical calculation without fitting parameters. Inset: Schematic of the energy barrier presented by a 20 crystal for proton and deuteron transfer. The solid black and blue lines indicate the zero point states for protons and deuterons, respectively. Credit: Dr. Marcelo Lozada-Hidalgo, University of Manchester. Isotope effect measured by mass spectrometry](https://scx1.b-cdn.net/csz/news/800a/2016/isotopeeffec.jpg)
The team's key insight derived from leveraging techniques developed in their previous paper1. "The most important was the ability to produce a large number of fully suspended one-atom-thick crystal membranes of graphene and hexagonal boron nitride, which allowed us to separate interfaces by a membrane that is only one atom thick. We're used to it in our lab now, but it's truly remarkable that graphene, an atomic scale crystal mesh, can sieve subatomic particles." Moreover, due to its atomic thinness, the researchers are able to probe into phenomena that were impossible to access before – and to do so at room temperature.
In their current paper, the scientists state that their approach offers a competitive and scalable way for hydrogen isotope enrichment. "Hydrogen, deuterium and tritium – the three isotopes of hydrogen – have very similar chemical properties, which makes them very hard to separate and requires some of the most energy-intensive processes in the chemical industry," Lozada-Hidalgo explains. "Until now, there was no direct separation method for hydrogen isotopes, so the solutions, while ingenious, were very expensive." He points out the technological implication of their results is that graphene and boron nitride are, in essence, extremely fine sieves – a finding that could have a large impact in, for example, removing tritium waste from water. This would be especially important in nuclear accidents like the Fukushima disaster, where while heavy radioactive waste such as uranium has been successfully removed, tritium, due to its similarity to hydrogen (and therefore to water) has so far proven particularly challenging to remove. In addition, because graphene is physically sieving the isotopes using only water at the input without additional chemical compounds, energy and process costs associated with this isotope separation method are lower than that of existing processes.
One of the great successes of the project, Lozada-Hidalgo says, was showing that macroscopic cracks and pinholes present in CVD graphene do not affect the approach's efficiency because hydrons (a collective name for the ions of all three hydrogen isotopes – that is, protons, deuteron and tritons) are electrochemically pumped only through the graphene areas that are electrically contacted. "We managed to scale devices to centimeter sizes, without which we would have gained significant insight into the proton transport process – but applications would still be a long way ahead. To do so, in one of our experiments we used graphene simultaneously as an electrode and a proton sieve – a completely new geometry possible only because of graphene's extraordinary properties." In this geometry the researchers used an electric current to pump protons through graphene, thereby sieving the isotopes.
"The really great feature of this geometry," he stresses, "is that it only pumps protons in the regions that have graphene because, quite simply, in the regions where graphene is absent there is no proton pump capability. This geometry is therefore very resilient towards cracks, which are bound to happen in real-life devices."
The study also showed that graphene is not the only material that can serve as an ionic sieve, and example being hexagonal boron nitride (hBN) monolayers. "Boron nitride is very attractive because it's an even better proton conductor than graphene, and would therefore allow for faster sieving." The team focused on graphene, Lozada-Hidalgo notes, because monolayer chemical vapor deposition (CVD) boron nitride is not yet commercially available in large quantities.
The scientists have other plans as well. "Next we want to work with tritium, Lozada-Hidalgo tells Phys.org, "since we couldn't use it in our previous experiments because it's radioactive. We're confident that we'll find the same results as with deuterium – but it's nevertheless interesting to demonstrate it." Beyond that, he adds, this being a very new field of research, they continue to discover new phenomena.
Even with this being a new discipline, there are other fields of research that already can be seen as potentially benefitting from the team's study. "Chemical engineering is an obvious field, but it's just one of them. Because hydrogen isotopes are used as tracers in chemical reactions, we believe our research could have very interesting implications in, for example, biology, where there is extensive research into the interaction of DNA and other biomolecules with graphene membranes. Chemistry is another example, in which conducting reactions with deuterium instead of hydrogen elucidates the limiting steps in chemical processes. Finally," Lozada-Hidalgo concludes, "there's a great deal to investigate using subatomically selective membranes – the ultimate selectivity a membrane can display – and two-dimensional crystals are the first membranes to show this capability. We're very excited about the possibilities down the road!"
More information: Sieving hydrogen isotopes through two-dimensional crystals, Science (2016) 351(6268): 68-70, doi:10.1126/science.aac9726
Related
1Proton transport through one-atom-thick crystals, Nature (2014) Nature 516: 227–230, doi:10.1038/nature14015
© 2016 Phys.org