Building a better salt trap: Scientists synthesize a molecular 'cage' to trap chloride
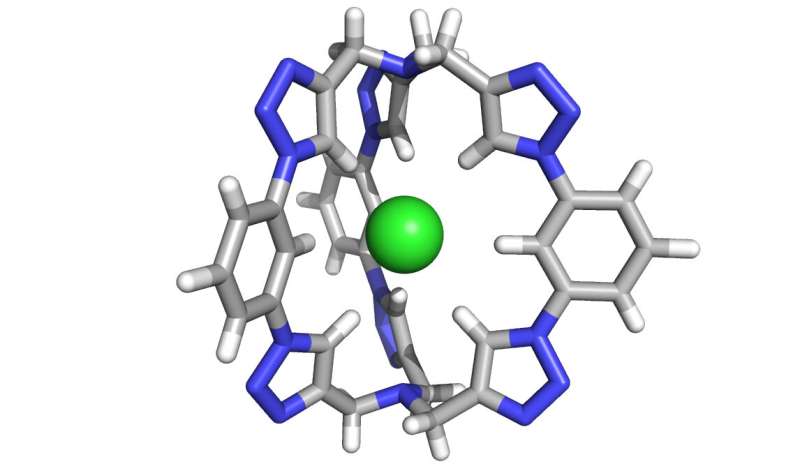
Indiana University researchers have created a powerful new molecule for the extraction of salt from liquid. The work has potential to help increase the amount of drinkable water on Earth.
Built using chemical bonds previously regarded as too weak, the new molecule is about ten billion times improved compared to a similar structure created over 10 years ago at IU. The molecules' design was reported May 23 in the journal Science.
"If you were to place one-millionth of a gram of this molecule in a metric ton of water, 100 percent of them will still be able to capture a salt," said Yun Liu, who led the study as a Ph.D. student in the lab of Amar Flood, the James F. Jackson Professor of Chemistry and Luther Dana Waterman Professor in the IU Bloomington College of Arts and Sciences' Department of Chemistry.
The molecule is designed to capture chloride, which is formed when the element chlorine pairs with another element to gain an electron. The most familiar chloride salt is sodium chloride, or common table salt. Other chloride salts are potassium chloride, calcium chloride and ammonium chloride.
At the same time that the human population continues to grow, the seepage of salt into fresh water systems is reducing access to drinkable water across the globe. In the U.S. alone, the U.S. Geological Survey estimates 271 metric tons of dissolved solids, including salts, enter freshwater streams per year. Contributing factors include the chemical processes involved in oil extraction, the use of road salts and water softeners, and the natural weathering of rock. It only takes one teaspoon of salt to permanently pollute five gallons of water.
The new salt-extraction molecule created at IU is comprised of six triazole "motifs"—five-membered rings comprised of nitrogen, carbon and hydrogen—which together form a three-dimensional "cage" perfectly shaped to trap chloride. In 2008, Flood's lab created a two-dimensional molecule, shaped like a flat donut, which used four triazoles. The two extra triazoles give the new molecule its three-dimensional shape, and ten billion-fold boost in efficacy.
The molecule is also unique because it binds chloride using carbon-hydrogen bonds, previously regarded as too weak to create stable interactions with chloride compared to the traditional use of nitrogen-hydrogen bonds. Despite expectations, the researchers found the use of triazoles created a cage so rigid as to form a vacuum in the center, which draws in chloride ions.

By contrast, cages with nitrogen-hydrogen bonds are often more flexible—and the vacuum-like center needed for chloride capture requires energy input, lowering their efficiency compared to a triazole-based cage.
"If you were to take our molecule and stack it up against other cages that use [stronger] bonds, we're talking many of orders of magnitude of performance increase," said Flood. "This study really shows that rigidity is underappreciated in the design of molecular cages."
The rigidity also enables the molecule to retain its shape after the central chloride has been lost, compared to other designs that collapse under the same circumstances due to their flexibility. This provides the molecule greater efficacy and versatility.
The work is also reproducible. The first molecule took nearly a year to synthesize, said Liu, who was shocked to discover the crystals required to confirm the molecule's unique structure formed after the experiment was left alone in the lab for several months—a surprising occurrence since this process typically requires careful monitoring.
The formation of the crystal represented a "eureka" moment, proving the molecule's unique design was actually viable. Later, Wei Zhao, a postdoctoral researcher in Flood's lab, was able to re-create the molecule in several months.
More information: "Chloride capture using a C–H hydrogen bonding cage" Science (2019). science.sciencemag.org/cgi/doi … 1126/science.aaw5145
Journal information: Science
Provided by Indiana University