May 6, 2019 feature
Engineering artificial cell membranes to drive in situ fibrin hydrogel formation
Re-engineering the cell membrane for improved biofunction is an emerging, powerful tool in cell biology to develop next-generation cell therapies. The process can allow users to supplement cells with added therapeutic functionalities. Additional functionalities can include cell homing, surface adhesion or resistance to hypoxia for enhanced cellular capabilities. However, the number of such examples on re-engineered plasma-membranes to activate membrane-bound enzymes that promote the assembly of extracellular matrix (ECM) proteins to promote cell functionalities are limited.
In a recent study, Robert C. Deller and co-workers at the interdisciplinary departments of Cellular and Molecular Medicine, Engineering, Functional Nanomaterials and Pharmacology, and Neuroscience in the UK, bioengineered a self-contained cell matrix-forming system. In the experiments, they modified the plasma membrane of human mesenchymal stem cells (hMSCs) to integrate a new thrombin construct, which gave rise to spontaneous fibrin hydrogel nucleation and growth when supplemented with human plasma concentration levels of fibrinogen in cell culture media.
The scientists bioengineered the cell membrane by synthesizing a membrane-binding supercationic thrombin-polymer surfactant complex. Thereafter, they observed cell differentiation in the resulting robust, stem cell-containing fibrin hydrogel constructs to form osteogenic and adipogenic cell lineages. The differentiated cells could eventually secrete fibrin to form self-supported bioengineered cellular monoliths that exhibited Young's moduli as expected of the native extracellular matrix. The results are now published in Nature Communications.
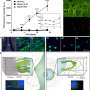
![Evaluating rhodamine (rh) labelled rh_thrombin, rh_sc_thrombin and [rh_sc_thrombin][ox890] on hMSC plasma membrane affinity. Cells labelled with CellMask (green) and rhodamine labelled thrombin (magenta) visualized with confocal microscopy. Video shows the rh_sc_thrombin [ox890] labelled hMSCs supplemented with fibrin gel conjugating with fibrinogen (green) to highlight fibrin formation emanating from the bioengineered cell surface. Credit: Nature Communications, doi: 10.1038/s41467-019-09763-0 Engineering artificial cell membranes to drive in situ fibrin hydrogel formation](https://scx1.b-cdn.net/csz/news/800a/2019/engineeringa.gif)
Advanced cell therapies are currently approaching clinical translation in response to an increasing demand for newly modified, cell-specific matrices (scaffolds) for biocompatible therapeutic performance. However, the rational design of matrices is extremely challenging since the cell phenotype and cell fate can be intertwined to a wide-range of scaffold-dependent factors; including cell adhesion, surrounding chemical composition, cell receptor stimulation, surface micro-/nano-morphology and mechanical stiffness. These factors immediately impact cells during in vitro tissue engineering, typically when cells are seeded to adhere on biocompatible and biodegradable scaffolds in the lab, where the scaffolds act as a surrogate extracellular matrix (ECM). Eventually, when the cells grow and differentiate, they can produce natural ECM of their own, to gradually replace the biomimetic scaffold material and form a structurally self-supporting biological entity.
A range of natural biocompatible polymers have produced such transient hydrogel scaffolds for tissue engineering; including chitosan, gelatin and fibrin. Fibrin hydrogels are the most popular among them, since they can be produced readily at room temperature using proteolytic cleavage. Biological fibrin formation occurs in response to injury, culminating from a biochemical cascade of proteolytic cleavage, which converts prothrombin to thrombin and forms a fibrin-hydrogel clot. Fibrin hydrogels can therefore mediate cellular biomolecular functions and regulate the osteogenic and chondrogenic differentiation of human stem cells such as hMSCs. They can also be conveniently delivered using syringes, albeit with complications related to reduced cell viability.
In the present work, Deller and co-workers first described a method to synthesize supercationic thrombin-polymer surfactant complexes that spontaneously bound to the plasma membrane of hMSCs to drive in situ fibrin hydrogel nucleation and growth. The resulting self-supporting hydrogel construct allowed high levels of metabolic activity as an artificial matrix for effective differentiation of stem cells to form adipogenic or osteogenic cell lineages. The scientists then showed the feasibility of the method of cell functionalization by injecting thrombin-labelled GFP-expressing fibroblasts (GFP: Green fluorescence protein) into a zebrafish (Danio rerio) skin wound model to demonstrate their in vivo biocompatibility for hemostatic applications.
![The synthesis and characterisation of the supercationic thrombin-polymer surfactant conjugate. a Schematic showing the electrostatic surface potential of native and supercationic thrombin (sc_thrombin) (PDB; 1UVS) at pH 7, highlighting the anionic (blue) and cationic (red) charged regions. Generation of the polymer surfactant corona (green halo) via electrostatic coupling of glycolic acid ethoxylate 4-nonylphenyl ether (ox890) to sc_thrombin gives [sc_thrombin][ox890]. b Zeta potential (ca. pH7; n = 3) of thrombin as a function of cationization times (0–120 min). Data reported as means ± standard deviation (s.d.). c Rate of fibrinogen solution (3.125 mg mL−1) gelation as measured by changes in turbidity (600 nm) catalysed by sc_thrombin subjected to various cationization times (0–120 min). Data shown as one-phase association curves of raw data. d MALDI-TOF MS spectra (m/z = 3) of native and sc_thrombin (60 min). Credit: Nature Communications, doi: 10.1038/s41467-019-09763-0 Engineering artificial cell membranes to drive in situ fibrin hydrogel formation](https://scx1.b-cdn.net/csz/news/800a/2019/2-engineeringa.jpg)
They synthesized the artificial membrane binding thrombin complex using a two-step process to generate an active supercationic thrombin construct (sc_thrombin). Deller et al. also generated a polymer surfactant corona or green halo using electrostatic coupling of glycolic acid ethoxylate 4-nonylphenyl ether (ox890) to sc_thrombin to engineer a third variant sc_thrombin [ox890].
The scientists controlled the reaction conditions (pH, temperature and chemical composition) carefully and monitored the reaction progress using zeta potentiometry across a period of two hours. They observed the activity of thrombin by monitoring/characterizing the increasing turbidity of the fibrinogen solution. They then obtained MALDI-TOF spectra (matrix-assisted laser desorption ionization time of flight mass spectroscopy) of the native and modified thrombin to show full cationization of the construct. When Deller et al. conducted compression testing of the resultant self-supporting structures, the Young's moduli were similar to soft fibrin hydrogels, indicating consistency.
To investigate thrombin adhesion to cell membranes, the scientists chose a monolayer of bone marrow derived hMSCs (with well-characterized adipogenic, chondrogenic and osteogenic pathways). First, they incubated a monolayer of hMSCs with fluorescently-labelled analogues of thrombin; thereafter, they labeled the hMSCs with a plasma membrane-specific dye and imaged immediately to confirm the thrombin-plasma membrane binding. Using time-lapse confocal fluorescence microscopy, they showed the nucleation and fibrin growth from the cells thereafter.
![3D projection of fibrin gel containing fibrinogen stained with Alexa 594 (red) fibrinogen and hMSCs incorporating sc_thrombin [ox890] stained with Hoeschst 33342, imaged after 60 min of cell associated fibrin formation in cell culture. Credit: Nature Communications, doi: 10.1038/s41467-019-09763-0. Engineering artificial cell membranes to drive in situ fibrin hydrogel formation](https://scx1.b-cdn.net/csz/news/800a/2019/1-engineeringa.gif)
Deller et al. also completed cell growth assays to determine relative metabolic activities of labelled hMSCs-thrombin to show the modified cells were without observable cytotoxicity in varying concentrations of thrombin (1 µm to 25 mM). Using confocal microscopy again, the scientists showed the arrangement of fibrin structures emanating from the plasma membrane of the hMSC monolayer, in contrast to hMSCs without thrombin. The work protocol thereby generated a 3-D fibrin hydrogel construct with dense cellular aggregates surrounded by a 3-D fibrin matrix.
The scientists also investigated the ability of the fibrin hydrogel system to sustain 3-D cultures for long-term growth; a requisite for tissue engineering, to show hMSC differentiation via adipogenic and osteogenic pathways. To verify the results, the scientists conducted extensive biomechanical tests on the cell types and tested for the upregulation of specific genes SOX9 and RUNX2 involved in chondrogenesis and osteogenesis respectively, using RT-PCR (reverse transcription polymerase chain reaction), to substantiate the fibrin hydrogel system sustained long-term hMSC proliferation in vitro.
After confirming the membrane re-engineering approach for in lab tissue engineering applications, Deller et al. investigated the ability to produce thrombin coated cells in lab for their injection at a site of injury to initiate a healing response for tissue-engineering applications in vivo. For this, the scientists used an in vivo Zebrafish model system to perform preliminary cell transplant studies. Zebrafish is a model organism, established for fluorescently labelled live cell imaging and thrombolytic and hemostatic processes; suited for the present work. The scientists isolated, labelled and delivered fluorescently labelled primary Zebrafish fibroblasts, labelled with sc_thrombin[ox890] conjugate via microinjection to show cell survival after 3 days at a site of incisional injury.
![In vivo zebrafish injury and [sc_thrombin][ox890] labelled GFP + fibroblast addition. Schematic representation of the in vivo adult zebrafish injury model. a Wildtype (non-transgenic) recipient zebrafish were anaesthetized and a 4 mm incisional injury made on the ventral upper thorax. A lateral view is shown. b Unlabelled or [sc_thrombin][ox890] labelled, FACS sorted GFP+ fibroblasts were injected at six sites around the edge of the incisional injury. At the desired time-point, fish were sacrificed and the tissue surrounding the incision was fixed, imaged and embedded for sectioning. A ventral view is shown. Ventral view of the area of tissue surrounding the incision at 3 dpi following transfer of c unlabelled or d [sc_thrombin][ox890] labelled GFP+ fibroblasts. Similar numbers of cells were retained at all wounds. The red line depicts the approximate position of the incisional injury which is fully re-epithelialized at this stage. Sections through the injury region at 3 dpi following transfer of e unlabelled or f [sc_thrombin][ox890] labelled GFP+ fibroblasts. No obvious differences were observed between wounds containing labelled or unlabelled cells. Arrows indicate the position of the incision. Credit: Nature Communications, doi: 10.1038/s41467-019-09763-0. Engineering artificial cell membranes to drive in situ fibrin hydrogel formation](https://scx1.b-cdn.net/csz/news/800a/2019/3-engineeringa.jpg)
Using macroscopic observations and histological assays, the scientists further revealed that there were no adverse effects between fish injected with engineered or native fibroblasts. However, Deller et al. expect to complete further investigations to understand the precise effects on the specific wound healing response using bioengineered cells in the future.
In this way, Deller et al. synthesized and characterized a new membrane active thrombin construct and demonstrated its application to drive in situ fibrin formation in the plasma membranes of stem cells. The scientists showed that thrombin-based fibrin hydrogel constructs prepared using the new protocol supported high levels of cell growth and viability to eventually produce a self-supporting tissue engineered construct. The stem cells were also able to differentiate along the well-defined adipogenic and osteogenic pathways while demonstrating Young's moduli similar to the native cells to indicate high levels of integration of the modifications. Deller et al. propose to optimize the protocols for further experiments in vitro prior to in vivo translation, to gain further insight to the enzymatic activity of cell membrane bound bioengineered proteins to develop biocompatible, hemostatic products.
More information: Robert C. Deller et al. Artificial cell membrane binding thrombin constructs drive in situ fibrin hydrogel formation, Nature Communications (2019). DOI: 10.1038/s41467-019-09763-0
D. E. Discher. Tissue Cells Feel and Respond to the Stiffness of Their Substrate, Science (2005). DOI: 10.1126/science.1116995
Tamer A.E. Ahmed et al. Fibrin: A Versatile Scaffold for Tissue Engineering Applications, Tissue Engineering Part B: Reviews (2008). DOI: 10.1089/ten.teb.2007.0435
Journal information: Nature Communications , Science
© 2019 Science X Network