April 26, 2019 feature
Stimulating the differentiation of bone precursors with organically modified hydroxyapatite (ormoHAP) nanospheres

Bioinspired materials mimic their natural counterparts for characteristic functionality in multidisciplinary applications forming a popular theme in biomaterials development. In bone tissue engineering, for instance, researchers focus on the natural composite architecture of bone, organically designed from complex structures of mineralized collagen. The resulting bioengineered constructs include inorganic/organic composites based on native mammalian bone components such as carbonated apatite and collagen. However, microparticle incorporation to material constructs can cause complications during premature in vivo resorbability, due to their brittle nature.
In a recent study, now published in Biomedical Materials, IOP Science, Christiane Heinemann and co-workers at the Max Bergmann Center of Biomaterials and Institute of Materials Science in Germany, engineered isolated nanospheres using organically modified hydroxyapatite (ormoHAP) – to form a composite scaffold aligning with preceding work of the same research team. Heinemann et al. engineered the new biomaterial using an electric field-assisted ion double migration process and embedded the nanospheres thus formed, in the foamed gelatin organic template, to form the composite scaffold.
The scientists tested the biodegradation rates of the biomaterials to show that they correlated with the degree of crosslinking (40%, 80%) conveyed during scaffold preparation and with the mineral content of the scaffolds (0%, 20%, 40%). They used a human cell co-culture model of osteoblasts and osteoclasts derived from bone marrow stromal cells and monocytes, to test the impact of ormoHAP-gelatin scaffolds on cell growth and differentiation for a period of 42 days.
The results confirmed that ormoHAP embedded in the gelatin matrix enhanced TRAP5b bioactivity (Tartrate-resistant acid phosphatase 5b); a group of enzymes synthesized in bone, followed by increased ALP activity (alkaline phosphatase, an osteoblast marker) and increased gene expression of BSPII (bone sialoprotein II – encoding a major structural protein of the bone matrix) in osteoblasts. The scientists proposed a sequence of cell cross-talk interactions, due to the presence and concentration of ormoHAP in the material, to explain the observed behavior in cell co-cultures in vitro.

The hydroxyapatite (HAP) nanocrystals self-assembled in the organic environment to form hollow spherical agglomerates in the experiments, which the scientists first characterized in depth due to their role in forming the bone substitute materials (BSM). Heinemann et al. chose gelatin as the underlying scaffold matrix material due to its compatibility with the electric field-assisted mineral formation process of nanospheres, while both constituents of the composite (HAP and gelatin) showed cytocompatibility during cell-material interactions as shown in previous studies in vitro.
Gelatin is a well-suited constituent to form bioinspired materials for bone tissue engineerng, as it is a denaturation product of collagen, with abundant availability, processability, biodegradation and low antigenicity; suited to develop new biomaterials. Materials scientists previously developed similar constructs as gelatin/alginate, gelatin/chitosan, gelatin/βTCP or gelatin/HAP composite scaffolds, where mineralized composites facilitated cell proliferation compared to monophasic substrates. In vitro experiments with co-cultures of diverse cell types are better suited to test biomaterials as they represent the natural conditions of intercellular interaction to simulate cell regeneration.
To more accurately replicate the conditions in vivo, Heinemann et al. previously conducted supplement-free co-cultures with osteoblasts and osteoclasts to test biomaterials during material-assisted bone regeneration. The work indicated the requirement of balanced crosstalk between bone-forming osteoblasts and bone-resorbing osteoclasts either via soluble factors or direct cell-cell contact, for efficient bone remodeling.
The scientists therefore unified the results of many previous studies in the present work, to determine the formation of bone-tissue like extracellular matrix deposits guided by the underlying biomaterial. Heinemann et al. co-cultured human bone marrow stromal cells (hBMSC) with human osteoblasts (hOB), and human monocytes (hMc) with human osteoclasts (hOC), without supplements on 3D composite (ormoHAP/Gelatin) scaffolds. They then conducted cell-material characterizations (tests) to investigate the influence of the organically modified HAP nanospheres (ormoHAP) on cell behavior and interactions in the lab.
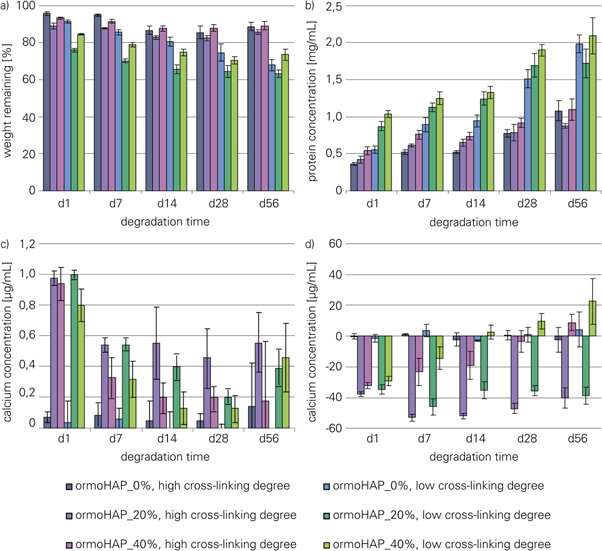
The scientists first engineered a variety of composites with ormoHAP embedded in gelatin to create multiple scaffolds for cell culture experiments, followed by testing them with scanning electron microscopy (SEM) images to understand the micro-/nano- architecture of the new material. They observed distinct surface patterning on the gelatin matrix due to homogenous ormoHAP distribution. Heinemann et al. produced a variety of such stable scaffolds on chemically crosslinked gelatin organic templates and tested their degradation behavior using buffer (phosphate buffered saline, PBS) or simulated body fluid (SBF) media to closely mimic in vivo biological conditions in the lab.
The scientists determined the effects of the percentage of gelatin cross-linking and the concentration of ormoHAP on the bioactivity and degradation of the new material, with comparative studies. In degradation studies with SBF or PBS, scaffolds with a lower degree of cross-linking degraded much faster, than those with higher cross-linking. By day 56, the scientists observed higher levels of bioactivity on scaffolds with 20 percent ormoHAP; determined by quantifying the levels of surface bound calcium. Although a concentration of 40 percent ormoHAP showed promising results initially, the values of surface-bound calcium decreased with time.
During co-culture experiments Heinemann et al. therefore compared two different concentrations of ormoHAP (20 percent and 40 percent), alongside scaffolds made of pure gelatin alone. The scientists strategically conducted cell culture studies from day 14 to day 28 and up until day 42, then using DNA analysis they quantified the cell nuclei and calculated the rate of cell proliferation to assess the total number of cells on the material surfaces, with no significant difference observed between the surfaces.
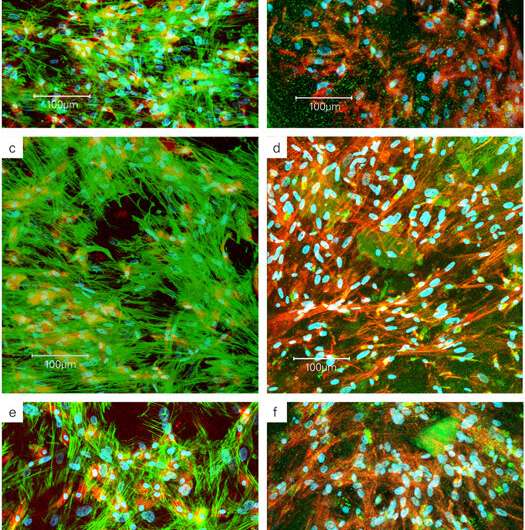
They quantified ALP activity, to assess osteogenic differentiation in monoculture and co-culture, which decreased after 14 days as cell maturation increased. To investigate the differentiation of hMc to hOB in co-culture, the scientists quantified TRAP5b activity, which remarkably increased with increasing ormoHAP content in the scaffold composition for material-assisted cell growth. By day 42 however the rates of enzyme activity decreased due to the limited lifespan of osteoclast cells. Heinemann et al. next conducted confocal laser scanning microscopy (cLSM) imaging to investigate co-culture interactions on the scaffold.
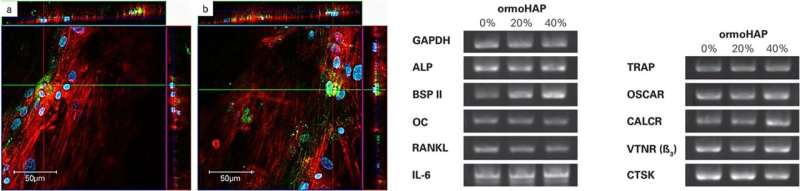
They observed the cell co-cultures displaying green actin skeleton, blue cell nuclei using fluorescent makers and used a red cell surface antigen marker (CD68) to detect the monocytes (hMc). Using microscopic images, the scientists observed varying cell morphology from spindle-shape to spherical-shape, detailing how cells interacted with the underlying new material. They detected TRAP, as brightly stained spots of green, increasingly concentrated within cells as the ormoHAP levels on the material surface increased, to highlight the effect of material-assisted cell growth. Heinemann et al. finally conducted gene analysis to determine the upregulation of specific markers related to cell differentiation using quantitative real-time polymerase chain reaction (qRT-PCR).
Notably they investigated BSPII (bone matrix encoding protein), RANKL (receptor activator of NF- κB ligand) involved in bone modeling/remodeling and the osteoclast maker OSCAR (osteoclast associated Ig-like receptor) that resorb bone - essential for bone homeostasis. The results indicated the upregulation of BPSII and OSCAR, verifying material-assisted cell differentiation in the present work.
In this way, Heinemann et al. extensively characterized cell-material interactions to understand the new, bioinspired ormoHAP materials during biofunctionalization . They showed the influence of the new scaffold-geometry on bone forming and resorbing cells, and on inter-cellular interactions with each other, using the cell co-culture study. The results will allow the scientist to achieve optimized production conditions to further improve and develop materials constructs for bioinspired materials engineering. The increased concentration of ormoHAP in the scaffolds stimulated cellular cross-talk between osteoblasts and osteoclasts as evidenced with specific markers of gene upregulation, with promising implications for further investigations of the new materials in bone tissue engineering.
More information: 1. Organically modified hydroxyapatite (ormoHAP) nanospheres stimulate the differentiation of osteoblast and osteoclast precursors: a co-culture study iopscience.iop.org/article/10. … 088/1748-605X/ab0fad Christiane Heinemann et al. 15 April 2019, Biomedical Materials, IOP Science.
2. Electric field-assisted formation of organically modified hydroxyapatite (ormoHAP) spheres in carboxymethylated gelatin gels www.ncbi.nlm.nih.gov/pubmed/27544814 Christiane Heinemann et al. October 2016, Acta Biomaterialia.
3. Stimulation of osteoblast responses to biomimetic nanocomposites of gelatin-hydroxyapatite for tissue engineering scaffolds www.ncbi.nlm.nih.gov/pubmed/15792549 Kim HW et al. September 2005, Biomaterials.
© 2019 Science X Network