September 14, 2018 feature
Advanced biomaterials with silk fibroin-bioactive glass to engineer patient-specific 3-D bone grafts
The complex architecture of bone is challenging to recreate in the lab. Therefore, advances in bone tissue engineering (BTE) aim to build patient-specific grafts that assist bone repair and trigger specific cell-signaling pathways. Materials scientists in regenerative medicine and BTE progressively develop new materials for active biological repair at a site of defect post-implantation to accelerate healing through bone biomimicry.
Rapidly initiation of new bone formation at the site of implantation is a highly desirable feature in BTE, and scientists are focused on fabricating grafts that strengthen the material-bone interface after implantation. Bioactive glass can bond with bone minutes after grafting, and silk fibroin, a natural fibrous protein has potential to induce bone regeneration. Hybrid materials that exploit these properties can combine the osteogenic potential and the load-bearing capacity for potential applications in large-load bone defect models.
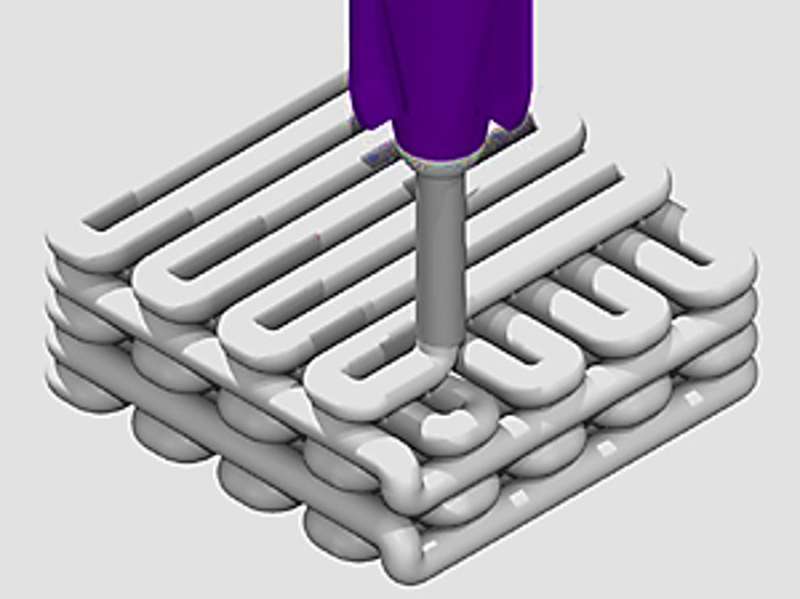
In a recent study, Swati Midha and co-workers developed a novel 3-D hybrid construct using silk-based inks with different bioactive glass compositions integrated to recreate a bone-mimetic microenvironment that supports osteogenic differentiation of bone marrow mesenchymal stem cell (BMSC) lines in the lab. Now published in Biomedical Materials, IOP Science, the scientists used direct writing instruments to produce the silk fibroin-gelatin-bioactive glass scaffolds (SF-G-BG). The results delivered appropriate cues to regulate the development of customized 3-D human bone constructs in vitro.
The authors explored two bioactive glass compositions (with and without strontium) ingrained in silk-based matrices. The work investigated (1) the mechanical properties of hybrid composites for their potential as inks to 3-D print scaffolds, followed by (2) the osteogenic potential of such SF-G-BG ink-based 3-D printed matrices, and (3) the underlying signaling mechanisms responsible for bone differentiation on 3-D printed constructs.
The ink was created using melt-derived glass, and after a series of optimizations with different concentrations, SF-G-BG composites optimized for printability and cytocompatibility were made possible. After printing, the 3-D SF-G-BG scaffolds were incubated in 80 percent ethanol to induce conformational changes in the constituting silk protein.

Physico-chemical properties of the materials were tested with FTIR and inductive coupled plasma mass spectrometry used to monitor the ion release profile of bioactive glasses within silk fibroin. Mesenchymal stem cells were cultured on the materials to understand mechanisms of cellular differentiation.
Typically, osteogenic differentiation on silk-based matrices is associated with Wnt/β signaling pathway activation, whereas bioactive glass activates a different set of osteogenic signaling pathways. Therefore, the authors investigated if these mechanisms of cellular differentiation were independent of each other or if cross-talk between them led to the induction of a new set of genes to regulate bone formation on the hybrid constructs.
In-depth molecular studies showed that SF-G-BG constructs containing Strontium (Sr) had superior osteogenic differentiation by driving mesenchymal stem cells towards osteoblastic and osteocytic phenotypes within 21 days of cell culture. Thereafter, the authors tested for the upregulation of six genes of interest to investigate osteoblastic differentiation, including the expression of Runt-related transcription factor (Runx2), a master gene that triggers the onset of osteogenic expression early in the cell differentiation cycle, to gradually decrease by day 7 as observed in the study.
Similarly, the authors tested for the upregulation of three specific genes expressed during osteocytic differentiation. Followed by studies to detect ionic release from bioactive glass in silk-gelatin ink constructs triggering bone morphogenetic protein 2 BMP-2, bone morphogenetic protein 4 BMP-4 and Indian hedgehog IHH cell signaling pathways that are critical to regulate bone formation in vivo. Gene ontology tests also determined the network of associated genes during osteogenic differentiation of BMSCs in 3-D printed SF-G-BG constructs.
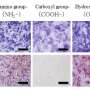
Most cells were detected to survive on the composite materials, confirming that SF-G-BG composition supported stem cell viability. The cell-material surfaces were observed with scanning electron microscopy (SEM) to visualize cell morphology and immuno-histochemical staining to visualize osteogenesis with specific antibodies. Genetic studies indicated that bioactive glass with Sr-ingrained silk fibroin scaffolds synergistically upregulated BMSC signaling pathways for enhanced differentiation and maturation, specifically activating the major signaling pathways (BMP-2, BMP-4 and IHH) critical to regulate bone formation in vivo. The results support further investigations in a preclinical animal model prior to engineering patient-specific 3-D SF-G-BG bone grafts in the lab.
More information: Swati Midha et al. Silk fibroin-bioactive glass based advanced biomaterials: towards patient-specific bone grafts, Biomedical Materials (2018). DOI: 10.1088/1748-605X/aad2a9
© 2018 Phys.org