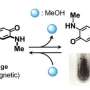
Turning a porous material's color on and off with acid

A research team led by Hokkaido University in Japan has constructed a porous material that is very stable and changes color when exposed to acid vapor. This is believed to be the first reported instance of a hydrogen-bonded organic framework changing color in response to acid. The findings are described in the Journal of the American Chemical Society.
Chemists are working to develop porous materials made from organic molecules that have structures with well-defined openings that can separate and store gasses. Such materials can also be used in electronic devices and sensors.
In particular, researchers are investigating how to make materials using molecules bonded together by hydrogen bonds, known as hydrogen-bonding organic frameworks (HOFs). HOFs are high-crystalline, flexible, and regenerable, which make them attractive candidates. But they can also be fragile and crumble apart.
Ichiro Hisaki, a chemist at Hokkaido University's Research Institute for Electronic Science, along with Anderrazzak Douhal, a photophysicist at University of Castilla La Mancha, Spain and their colleagues developed a hexagon-shaped framework, called CPHATN-1a, and found a surprising attribute – it changes color from yellow to reddish-brown when exposed to acid solution or acid vapor. When the acid solution or vapor is removed, either through heating or ambient evaporation, the HOF reverted back to its original yellow color.

The researchers determined that the color change is caused by protons adding onto nitrogen atoms within the compound, which shifts the spectrum of light absorbed.
Additional tests revealed that CPHATN-1a is extremely stable, maintaining its porous structure at temperatures to at least 633 Kelvin (359 C). The robust material also stood up to heated, common organic solvents, including chloroform, ethanol and water, retaining its structure rather than dissolving or breaking apart.
"The present results would open a door to develop new porous materials with stimuli responsiveness," the researchers note. "These could be used in the creation of new sensors or towards the visualization of minute chemical reactions."
More information: Ichiro Hisaki et al. Acid Responsive Hydrogen-Bonded Organic Frameworks, Journal of the American Chemical Society (2019). DOI: 10.1021/jacs.8b12124
Journal information: Journal of the American Chemical Society
Provided by Hokkaido University