Scientists design new responsive porous material inspired by proteins
Scientists from the University of Liverpool have, for the first time, synthesized a new material that exhibits structural change and triggered chemical activity like a protein.
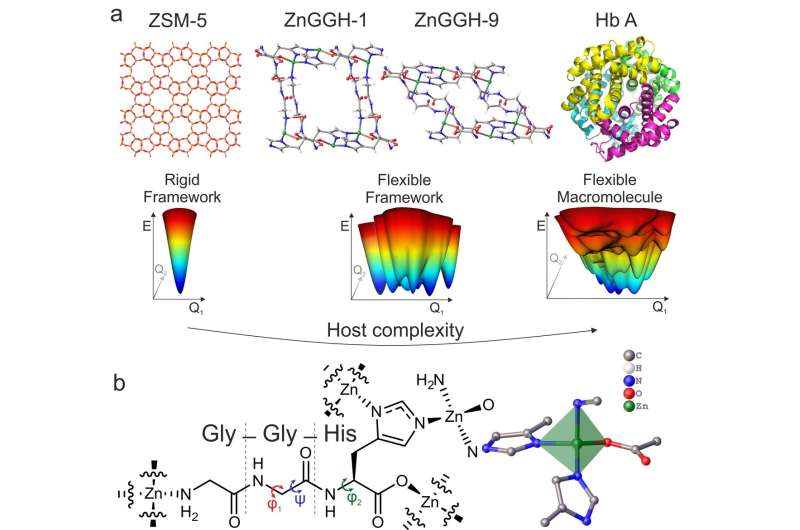
In research published in the journal Nature, Liverpool scientists produced a flexible crystalline porous material with small pores (< 1 nanometre) composed of metal ions and small peptide molecules that can change its structure in response to its environment to carry out specific chemical processes.
Porous materials are widely used in industry as catalysts for the production of fuels and chemicals and in environmental remediation technologies as adsorbers for the removal of harmful compounds from air and water.
These materials are rigid, with just one structure, unlike the proteins used by living systems to perform chemistry.
Proteins can change their structures to carry out chemical processes in response to their environment.
Like a protein, the new porous material can adopt multiple structures, and it can be controllably transformed from one structure to another by changes in its chemical environment. This allows it to perform a chemical process, such as taking up a particular molecule from its surroundings, in response to an imposed change in the surrounding solution.

Professor Matt Rosseinsky, who led the research, said: "These porous materials use the same atomic-scale mechanisms as proteins to switch between structures, which gives us the opportunity to develop new ways to manipulate and change molecules with synthetic materials that are inspired by biology.
"This offers exciting scientific possibilities, for example in catalysis, through the design of materials that can dynamically select the structure needed for a particular task."
The research team applied a combination of experimental and computational techniques to reveal the principles of the structural flexibility and activity of this new material.
They are now working on the development of the next generation of functional flexible porous materials whose performance is controlled by the changes in structure in response to changes in the chemistry surrounding them.
More information: Alexandros P. Katsoulidis et al, Chemical control of structure and guest uptake by a conformationally mobile porous material, Nature (2019). DOI: 10.1038/s41586-018-0820-9
Journal information: Nature
Provided by University of Liverpool