Modular construction—on a molecular scale
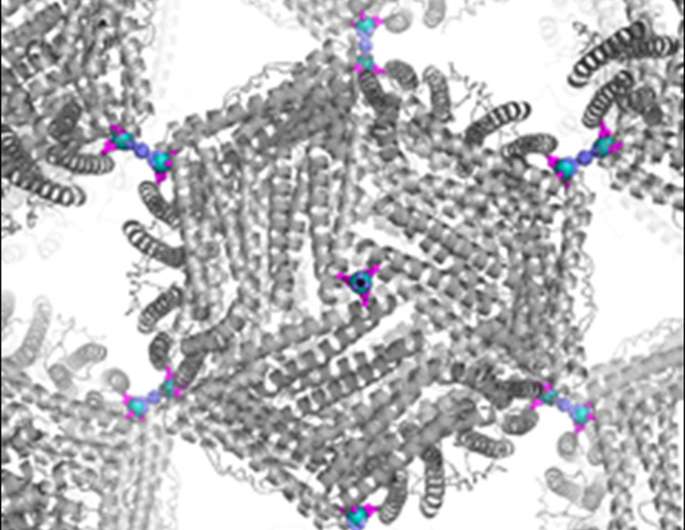
Modular constructions from cages (proteins), hubs (metal ions), and struts (organic linkers) allows the rational design of porous scaffolds. The inherent chemical and structural diversity of these building blocks leads to a new class of versatile, self-assembled materials.
This is the first instance where synthesis of a crystalline framework in which proteins as well as metal ions and organic molecules are essential construction components. This new fabrication route to functional materials has potential applications such as hydrogen fuel storage and carbon capture.
Formation of a 3-dimensional (3D) porous, crystalline framework material—formed by spherical proteins that assemble via directed interactions between these proteins, metal ions, and organic linkers—has been realized for the first time. Proteins hold great potential as building blocks for crystalline materials due to their wide range of available chemical and structural motifs combined with exquisite capabilities for catalysis and electron transfer. However, successful design and synthesis of 3D protein crystals is rare. Considerably more success in synthesizing crystalline materials has been achieved using small organic and inorganic building blocks, particularly the versatile metal-organic frameworks (MOFs) for purification and catalytic applications.
Researchers at the University of California, San Diego engineered a spherical, cage-like protein (ferritin) to display eight zinc ions on its surface in a symmetrical fashion. These surface zinc ions were analogous to small metal hubs in MOFs. Bifunctional organic linkers then bridged the zinc ions between two proteins, causing the nanoscale metal hubs to self-assemble into a predicted 3D crystalline lattice arrangement. The resulting crystals were highly porous, had very high solvent content, and allowed the proteins to perform their native enzymatic activity (transforming soluble iron ions into crystalline iron oxides). This new self-assembly strategy could lead to new materials for catalysis and separations.
More information: Pamela A. Sontz et al. A Metal Organic Framework with Spherical Protein Nodes: Rational Chemical Design of 3D Protein Crystals, Journal of the American Chemical Society (2015). DOI: 10.1021/jacs.5b07463
Journal information: Journal of the American Chemical Society
Provided by US Department of Energy



















