Removing toxic mercury from contaminated water
Water contaminated with mercury and other toxic heavy metals is a major cause of environmental damage and health problems worldwide. Now, researchers from Chalmers University of Technology, Sweden, present a totally new way to clean contaminated water through an electrochemical process. The results are published in the scientific journal Nature Communications.
"Our results have really exceeded the expectations we had when we started with the technique," says the research leader Björn Wickman, from Chalmers' Department of Physics. "Our new method makes it possible to reduce the mercury content in a liquid by more than 99 percent. This can bring the water well within the margins for safe human consumption."
According to the World Health Organisation (WHO), mercury is one the most harmful substances for human health. It can influence the nervous system, the development of the brain, and more. It is particularly harmful for children and can also be transmitted from a mother to a child during pregnancy. Furthermore, mercury spreads very easily through nature, and can enter the food chain. Freshwater fish, for example, often contain high levels of mercury.
In the last two years, Björn Wickman and Cristian Tunsu, researcher at the Department of Chemistry and Chemical Engineering at Chalmers, have studied an electrochemical process for cleaning mercury from water. Their method works via extracting the heavy metal ions from water by encouraging them to form an alloy with another metal.
"Today, cleaning away the low, yet harmful, levels of mercury from large amounts of water is a major challenge. Industries need better methods to reduce the risk of mercury being released in nature," says Björn Wickman.
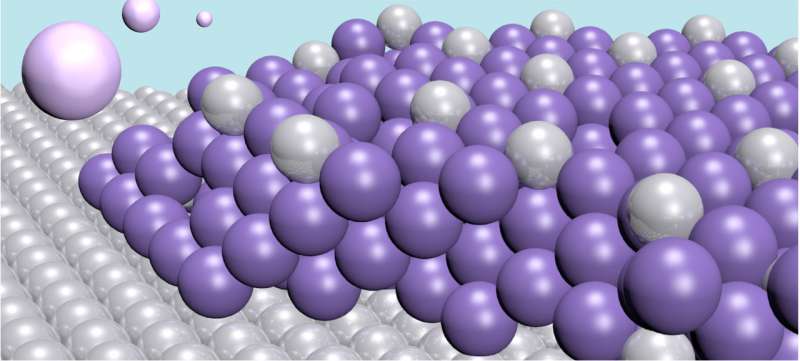
Their new method involves a metal plate—an electrode—that binds specific heavy metals to it. The electrode is made of the noble metal platinum, and through an electrochemical process it draws the toxic mercury out of the water to form an alloy of the two. In this way, the water is cleaned of the mercury contamination. The alloy formed by the two metals is very stable, so there is no risk of the mercury re-entering the water.
"An alloy of this type has been made before, but with a totally different purpose in mind. This is the first time the technique with electrochemical alloying has been used for decontamination purposes," says Cristian Tunsu.
One strength of the new cleaning technique is that the electrode has a very high capacity. Each platinum atom can bond with four mercury atoms. Furthermore, the mercury atoms do not only bond on the surface, but also penetrate deeper into the material, creating thick layers. This means the electrode can be used for a long time. After use, it can be emptied in a controlled way. Thereby, the electrode can be recycled, and the mercury disposed of in a safe way. A further positive for this process is that it is very energy efficient.
"Another great thing with our technique is that it is very selective. Even though there may be many different types of substance in the water, it just removes the mercury. Therefore, the electrode doesn't waste capacity by unnecessarily taking away other substances from the water," says Björn Wickman.

Patenting for the new method is being sought, and in order to commercialise the discovery, the new company Atium has been set up. The new innovation has already been bestowed with a number of prizes and awards, both in Sweden and internationally. The research and the colleagues in the company have also had a strong response from industry.
"We have already had positive interactions with a number of interested parties, who are keen to test the method. Right now, we are working on a prototype which can be tested outside the lab under real-world conditions."
Potential uses for the new method
The technique could be used to reduce the amount of waste and increase the purity of waste and process water in the chemical and mining industries, and in metal production. It can contribute to better environmental cleaning of places with contaminated land and water sources.
It can even be used to clean drinking water in badly affected environments because, thanks to its low energy use, it can be powered totally by solar cells. Therefore, it can be developed into a mobile and reusable water cleaning technology.
More on heavy metals in our environment
Heavy metals in water sources create enormous environmental problems and influence the health of millions of people around the world. Heavy metals are toxic for all living organisms in the food chain. According to the WHO, mercury is one of the most dangerous substances for human health, influencing our nervous system, brain development and more. The substance is especially dangerous for children and unborn babies.
Today there are strict regulations concerning the management of toxic heavy metals to hinder their spread in nature. But there are many places worldwide which are already contaminated, and they can be transported in rain or in the air. This results in certain environments where heavy metals can become abundant, for example fish in freshwater sources.
In industries where heavy metals are used, there is a need for better methods of recycling, cleaning and decontamination of the affected water.
More information: Cristian Tunsu et al, Effective removal of mercury from aqueous streams via electrochemical alloy formation on platinum, Nature Communications (2018). DOI: 10.1038/s41467-018-07300-z
Journal information: Nature Communications
Provided by Chalmers University of Technology