High-efficiency, low-cost catalyst for water electrolysis
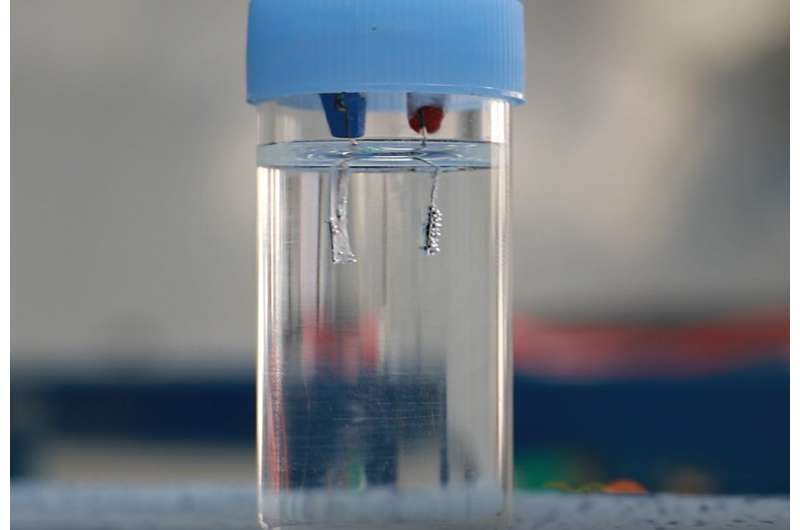
A research team led by Professor Sangaraju Shanmugam of Energy Science and Engineering at DGIST has developed highly efficient, ultra-durable core-shell nanostructured electrocatalyst and successfully replaced the precious anode in water electrolysis, through the collaboration with the research group of Pacific Northwest National Laboratory (PNNL).
The replacement of conventional fuels with renewable energy resources is a suitable approach to achieving an eco-friendly environment and decreasing future energy demands. Thus, electrochemical energy generation or conversion in renewable energy devices, which depends on anode and cathode reactions, has received much attention.
In electrocatalytic water splitting, oxygen gas generates in the anode due to the oxygen evolution reaction (OER), a slow electrochemical reaction as compared with the hydrogen evolution reaction (HER). Thus, a suitable electrocatalyst is needed for stable electrocatalytic water splitting.
Development of efficient, durable, low-cost OER electrocatalysts is important for water electrolyzer energy devices. Until now, the ruthenium and iridium oxides were considered state-of-the-art electrocatalysts in OER, but the lack of stability limits their use in large-scale water splitting, hindering widespread commercialization.
Professor Shanmugam's team, along with researchers from PNNL, have focused on developing an alternative low-cost, non-noble metal electrocatalyst to replace the noble metal anode electrode in efficient water splitting. Carbon-supported metal is considered an efficient electrocatalytic material for enhanced OER in water splitting. So far, most of the developed electrocatalysts have featured higher carbon content and less metal active specious content. The higher carbon amount mired the real metal active sites, and resulted in a faster carbon corrosion conditions. This led to lower electrocatalytic activity.
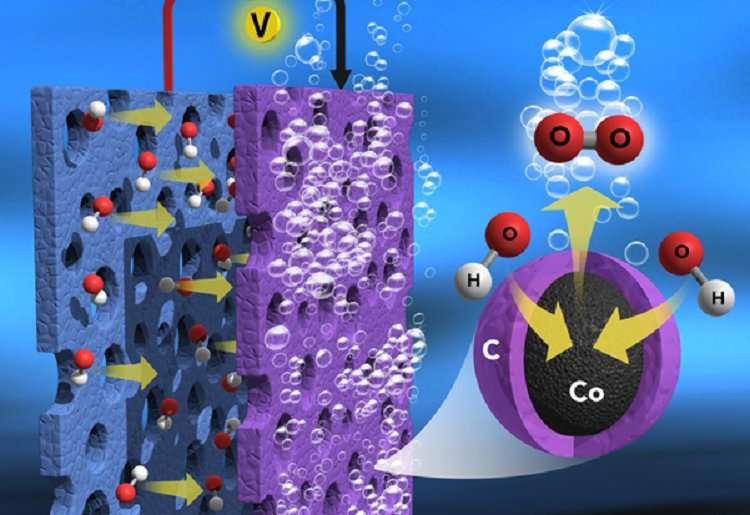
In the study, the researchers found that a large number of inorganic cobalt metal ions bridged by organic ligands in the Prussian blue are a suitable precursor for developing ultra-stable, metal-rich, nitrogen-doped graphitic nanocarbon-encapsulated core-shell electrocatalysts for the sluggish OER (anode) in water splitting.
When heated (600 to 900 degrees C) in an inert atmosphere, the cobalt metal ions and organic ligands in the salt are transformed into cobalt metal and nitrogen-doped graphitic thin carbon layers, respectively, which form the thin carbon layer, encapsulated metallic, cobalt core-shell nanostructures (Core-Shell Co@NC). The thin carbon layers have a strong interaction with cobalt metal, which promote less carbon corrosion, exhibit excellent electron movement, and have more cobalt metal exposure to the reaction medium, including the formation of nanosized morphology without particle aggregation.
The combined effect of carbon and cobalt metal in the electrodes achieves more efficient electrocatalytic OER activity than precious metal electrodes for efficient water splitting. Therefore, the non-noble metal-rich electrode is an alternative, active, stable, and less expensive OER anode for cost-effective H2 gas production in commercial-scale water electrolysis.
"We anticipate this to be a unique approach to developing metal-rich, reduced-carbon composite nanostructures that have enhanced metal active sites, which feature thin carbon layer protection and ultra-fast electron movement in the catalyst surface, that will enhance the electrochemical activity and stability of electrocatalysts," says Professor Shanmugam. "We will carry out the follow-up studies that can be used to understand the real OER mechanism on the active species in the presence of nanocarbon coating."
This research outcome was published in the online edition of Advanced Energy Materials on 11th of January 2018, a reputed international journal in the field emerging materials.
Interview with Professor Sangaraju Shanmugam (Department of Energy Science & Engineering):
Q. What are the differences compare to previous studies?
A. In the previous studies, the researchers prepared the carbon coated metals from various precursors, including metal-organic frameworks (MOFs). The obtained catalysts exhibit more carbon with reduced graphitic nature, and the carbon covered the active metal sites. Thus, most of the active metal sites are not utilized properly by the electrochemical reactions. Also, due to the substantial carbon corrosion, those catalysts are not suitable enough for the sluggish OER in water splitting at the higher positive potential with lack of instability in harsh electrolyte conditions. Accordingly, in this work, we prepared the metal-rich, thin nanocarbon (NC) layers encapsulated electrocatalyst of core-shell Co@NC nanostructures from a single precursor Prussian blue (PB) analogue. The Co@NC showed enhanced oxygen evolution activity and ultrastability on the current collector of nickel foam. Overall, the thin and uniform carbon layers provide the fast electron movements, more metal active sites utilization with easy electrolyte penetration. Most importantly, it can protect the active metal sites from the corrosion with minimal exposing and also the strong interaction between metal and carbon layers exhibits the synergistic effect towards the excellent activity and ultra-stability (over 350 h) of core-shell Co@NC nanostructures with less possibility of carbon oxidation.
Q. How can it be utilized?
A. Based on the remarkable OER performance, kinetics and long-term stability of core-shell Co@NC nanostructures as compared to the state-of-the-art Noble metal based electrocatalysts, such as IrO2 and RuO2, it is the most suitable candidate to replace precious metal OER electrodes for reducing the overall cost of the water electrolyzer system. Thus, the development of efficient and durable non-noble metal electrocatalyst in water electrolyzer is the main obstacle for successful commercialization of water electrolyzers.
Q. How long will it be required for commercialization?
A. The process is readily available for the fabrication of cost-effective catalysts. But we still have to evaluate the integration of this catalytic system in a polymer electrolyte membrane electrolyzer .Studies are underway to understand the OER mechanism on this electrocatalyst. So for commercialization, it may require a year with complete understanding of activity and stability.
Q. What are the challenges for commercialization?
A. We have to make the uniform coating of this catalyst on the larger size current collectors without any peeling. So we need to find a more suitable coating methodology. Also, as with precious OER electrocatalysts, we have to understand the precise OER mechanism on this electrocatalysts to maintain/avoid activity losses due to the unwanted side reactions, etc.
Q. What is the motivation for your research?
A. The primary motivation of this work is to replace the precious anode in water electrolyzer systems with high activity and stability. So to improve the activity and stability, we tried to introduce the very thin carbon coating on the metal active sites. Overall, the development metal-rich and carbon less OER electrocatalysts with proper utilization of metal-active species and metal-carbon synergistic effect to overcome the sluggish anode reaction in water electrolysis.
Q. What is the final goal you would like to achieve through this research?
A. Based on this research, we understand that the metal-rich electrocatalysts are among the most suitable materials for excellent OER activity. So we want to prepare the cheapest anode electrocatalysts by using the same methodology and eliminate the use of precious electrodes in the water electrolyzer system for the production of green and sustainable hydrogen in large scale.
More information: Arumugam Sivanantham et al, A Stable Graphitic, Nanocarbon-Encapsulated, Cobalt-Rich Core-Shell Electrocatalyst as an Oxygen Electrode in a Water Electrolyzer, Advanced Energy Materials (2018). DOI: 10.1002/aenm.201702838
Journal information: Advanced Energy Materials
Provided by DGIST