Unlocking the potential of metal nanoparticles as catalysts for fast and efficient CO2 conversion

O2 and sustainably produced hydrogen have the potential to serve as ingredients for converting electrical power generated by windmills or solar panels into a gas fuel. This 'power-to-gas' concept can solve two problems at once, reducing CO2 emissions while creating more flexible applications of sustainable energy. However, profitable conversion of CO2 would require an extremely effective catalyst. Researchers from Utrecht University have found a way to study the conversion process in detail and to determine the perfect size for the catalytic nickel nanoparticles. The researchers will publish their results in Nature Catalysis on Monday, 29 January.
Lead author Charlotte Vogt says, "When we make metal nanoparticles smaller and smaller, they start to show very different properties to what we expect and understand from classical physics and chemistry." Together with colleagues Florian Meirer and Bert Weckhuysen from Utrecht University and researchers from chemical company BASF, Vogt found that nickel particles exhibit optimal catalytic activity at a size of 2.5 nanometres, about 40,000 times smaller than a human hair. The researchers also found that a specific architecture of these tiny nickel particles facilitates the activation of CO2.
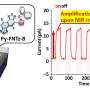
In order to understand how these nickel nanoparticles behave during the conversion of CO2, the researchers studied the catalysts in action. In cooperation with scientists at the Swiss Light Source in Switzerland, they developed an ultra-fast measuring tool to study their catalysts at work. This allowed the researchers to unlock the mechanism behind the CO2 conversion process in great detail. Thus, they identified both formate and adsorbed carbon monoxide as reaction intermediates.
The project involved a close collaboration between researchers at BASF, Utrecht University, Lehigh University in the United States, and the synchrotron facilities at the Paul Scherrer Institute in Switzerland. "This collaboration has brought us a better understanding of how these solid catalysts work, putting us in the position to unlock the true potential of small metal nanoparticles for CO2 catalysis," says Bert Weckhuysen, Professor of Inorganic Chemistry and Catalysis at Utrecht University.
More information: Unraveling Structure Sensitivity in CO2 Hydrogenation over Nickel, Charlotte Vogt, Esther Groeneveld, Gerda Kamsma, Maarten Nachtegaal, Li Lu, Christopher J. Kiely, Peter H. Berben, Florian Meirer, Bert M. Weckhuysen, Nature Catalysis, 29 January, DOI: 10.1038/s41929-017-0016-y
Journal information: Nature Catalysis
Provided by Utrecht University Faculty of Science