Long-lived catalytic converter reduces diesel emissions
Diesel vehicles today emit fewer pollutants than older vehicles, thanks to a zeolite catalytic converter that was invented around 10 years ago. Although many groups have investigated this catalyst since then, it was still unclear why it is so much more effective. By observing the catalyst's interior in three dimensions at the nanoscale level, researchers from Utrecht University and Oak Ridge National Laboratory (ORNL) in the U.S. have now found the explanation. After simulating 217,000 km of wear, they compared a 'new' and an 'old' version of the catalyst in detail. This revealed the current catalyst retains much more of its 'younger' structure than previously used diesel catalysts. The researchers also found the underlying reasons that this catalyst is so much more stable over its lifespan, and experiences only minimal damage compared to previously used catalysts. The results are published today in Nature Communications.
Diesel catalytic converters are exposed to frequent temperature changes, extremely hot steam and pollutants, but they must remain stable for the entire life of the vehicle. That stability is due in part to the complexity of the catalyst. "At first glance, zeolites may seem easy to understand, but the more you study them, the more fascinated you become by their complexity," says Joel Schmidt (Utrecht University), the publication's lead author. "This complexity is what makes zeolite catalytic converters so effective, but it also makes them exceptionally difficult to understand."
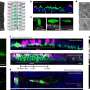
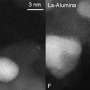
Schmidt and his colleagues have analysed the complexity of the zeolite catalyst using a unique and powerful characterization method called atom probe tomography. They could visualise all of the catalyst's relevant chemical elements in 3-D at nanoscale resolution, before and after a 217,000 km simulated aging procedure.
The researchers found that after this aging process, the current catalyst retains much more of its 'younger' structure than previously used diesel catalysts. They also discovered that this was because its structural properties prevent the formation of a deactivating copper aluminate oxide phase. Thus, the optimal nanoscale distribution of elements within the catalyst responsible for the clean combustion process remains intact.
Cleaner air
"With our approach, we were able to add another piece to the puzzle of how to design catalysts that perform just as well at the end of a vehicle's life as they did the day they rolled out of the factory," says Professor Bert Weckhuysen, co-author of the publication. "Since zeolite catalysts are used broadly in the chemical industry as well, insight on the migration of chemical elements under catalytic operation conditions is a very relevant contribution to realise more sustainable processes."
More information: Joel E. Schmidt et al, Nanoscale tomography reveals the deactivation of automotive copper-exchanged zeolite catalysts, Nature Communications (2017). DOI: 10.1038/s41467-017-01765-0
Journal information: Nature Communications
Provided by Utrecht University Faculty of Science