Utrecht chemists prove Nobel Prize-winner Olah correct
In 1877, Charles Friedel and James Craft discovered a chemical reaction for quickly producing raw materials for plastics, fine chemicals and detergents. More than 100 years later, in 1994, the American George Olah won the Nobel Prize for Chemistry for understanding the mechanism behind this important reaction. This is also how it has been described in Chemistry textbooks for almost 30 years. However, recently, some chemists claimed the Nobel Prize-winning reaction mechanism doesn't always apply. Now, chemists at Utrecht University refute this claim. They managed to detect the two most important reaction intermediates from Olah's reaction mechanism in the situation under debate. Olah, who passed away in March, has now received posthumous confirmation.
The results of the study were published online in Nature Catalysis on 20 November.
The reaction intermediates of this process are known as the Wheland complex and the pi complex. These molecular fragments have a short life, as they are quickly converted into the next reaction intermediate in the process or into the end product. In order to understand this process, or more importantly, to control it, it is essential to know whether the reaction progresses through these reaction intermediates.
Green route to polystyrene
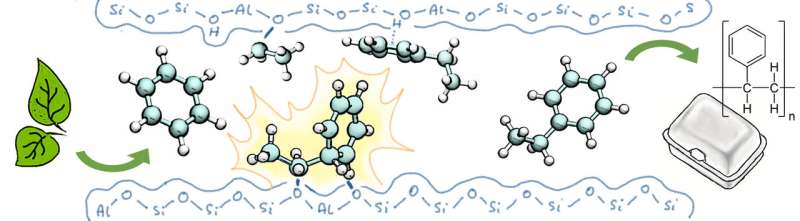
The reaction intermediates were proven in a study of a 'green' route for producing the commonly used plastic polystyrene. "Although it wasn't the primary goal of our research, it was still awesome that we were able to confirm Olah's proposal for the reaction mechanism," says first author Abhishek Dutta Chowdhury. "But the other results were interesting, as well. If we want to use less raw fossil material, then it is essential that we can understand how these kinds of important industrial processes occur at the molecular level."
Styrene, the foundation for polystyrene, is produced at an industrial scale using the Friedel-Craft reaction of benzene and ethylene derived from crude oil. The green route uses biomass and bioethanol instead, but the reaction is the same. One crucial element of this process is the development of an optimal catalyst that can ensure that the reaction is conducted quickly, effectively and at the ideal temperature and pressure. In this case, the catalyst is an extremely porous material called a zeolite.
Advanced characterization
In order to understand how the reaction process and the catalyst can be optimised, the researchers followed the reaction using advanced characterization techniques. With operando spectroscopy, they were able to observe the reaction in the pores of the zeolite in real time. Advanced multidimensional solid-state NMR methods, initially developed for applications to biomolecules, allowed the researchers to characterize the molecular structure of the formed products and intermediates, as well as their mobility and interaction with the catalyst material.
The results of their study left no doubt: the reaction occurred exactly as George Olah proposed in the diagram below. "That makes our study an excellent example of how socially relevant scientific research can contribute to our fundamental knowledge of that same science," says Prof Bert Weckhuysen of Utrecht University, who led the research project together with his fellow Professor Marc Baldus.
More information: Electrophilic aromatic substitution over zeolites generates Wheland-type reaction intermediates. Abhishek Dutta Chowdhury, Klaartje Houben, Gareth T. Whiting, Sang-Ho Chung, Marc Baldus and Bert M. Weckhuysen, Nature Catalysis 2018, 1, DOI: 10.1038/s41929-017-0002-4
Provided by Utrecht University Faculty of Science